-Cellstain- Calcein-AM

Live Cell Staining
-
Product codeC326 -Cellstain- Calcein-AM
-
CAS No.148504-34-1
-
Chemical name3',6'-Di(O-acetyl)-4',5'-bis[N,N-bis(carboxymethyl)aminomethyl]fluorescein, tetraacetoxymethyl ester
-
MWC46H46N2O23=994.86
| Unit size | Price | Item Code |
|---|---|---|
| 1 mg | C326-10 |
Description
Calcein-AM readily passes through the cell membrane of viable cells because of its enhanced hydrophobicity compared to Calcein. After Calcein-AM permeates into the cytoplasm, it is hydrolyzed by esterases to Calcein, which remains inside the cell (Fig. 1). Among other reagents, including BCECF-AM and Carboxy-fluorescein diacetate, Calcein-AM is the most suitable fluorescent probe for staining viable cells because of its low cytotoxicity. Calcein does not inhibit any cellular functions such as proliferation or chemotaxis of lymophocyte. In addition, viability assays using Calcein are reliable and correlate well with the standard 51Cr-release assay. The excitation and emission wavelengths of calcein are 490 nm and 515 nm, respectively.
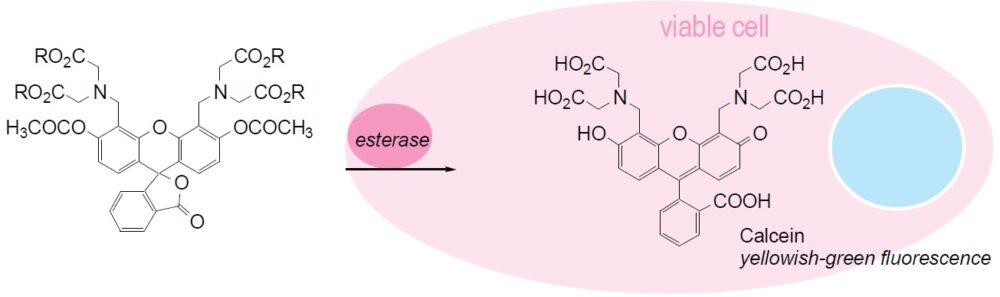
Fig. 1 Cell staining mechanism
Technical info
*caution*
Please tap the tube before opening, and open it with care. The content may have relocated from the bottom of the tube during the shipping.

Staining Procedure
1.Prepare 1 mM Calcein-AM solution with DMSO and dilute to prepare 1-50 μM Calcein-AM solution with PBS.a) 2.Add Calcein-AM solution with 1/10 of the volume of cell culture medium to the cell culture.b) 3.Incubate the cell at 37ºC for 15-30 min. 4.Wash cells twice with PBS or an appropriate buffer. 5.Observe the cells under a fluorescence microscope with 490 nm excitation and 515 nm emission filters.
a) If the Calcein-AM has difficulty loading into cells, use a detergent such as Pluronic F127.
b) Or you may replace the culture medium with 1/10 concentration of Calcein-AM buffer solution.
Data
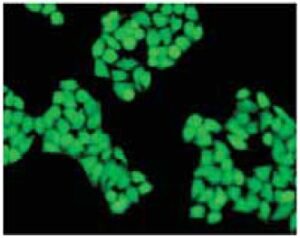
Fig. 2 Cell staining with Calcein-AM Cell type: HeLa
References
1. K. McGinnes, et al., A Fluorescence NK Assay Using Flow Cytometry. J Immunol Methods. 1986;86:7-15.
2. S. J. Morris, Real-time Multi-wavelength Fluorescence Imaging of Living Cells. BioTechniques. 1990;8:296-308.
3. S. A. Weston, et al., New Fluorescent Dyes for Lymphocyte Migration Studies Analysis by Flow Cytometry and Fluorescent Microscopy. J Immunol Methods. 1990;133:87-97.
4. D. M. Callewaert, et al., Characterization of Effector-Target Conjugates for Cloned Human Natural Killer and Human Lymphokine Activated Killer Cells by Flow Cytometry. Cytometry. 1991;12:666-676.
5. H. Xie, et al., Intercellular Communication Through Gap Junctions Is Reduced in Senescent Cell. Biophys J. 1992;62:45-47.
6. S. A. Weston, et al., Calcein: a Novel Marker for Lymphocytes Which Enter Lymph Nodes. Cytometry. 1992;13:739-749.
7. X. M. Wang, et al., A New Microcellular Cytotoxicity Test Based on Calcein AM Release. Hum Immunol. 1993;37:264-270.
8. N. G. Papadopoulos, et al., An Improved Fluorescence Assay for the Determination of Lymphocyte-Mediated Cytotoxicity Using Flow Cytometry. J Immunol Methods. 1994;177:101-111.
9. L. S. D. Clerck, et al., Use of Fluorescent Dyes in the Determination of Adherence of Human Leucocytes to Endothelial Cells and the Effects of Fluorochromes on Cellular Function. J Immunol Methods. 1994;172:115-124.
10. H. Ohata, et al., Confocal Imaging Analysis of ATP-Induced Ca2+ Response in Individual Endothelial Cells of the Artery in Situ. Am J Physiol. 1997;272:C1980-C1987.
Handling and storage condition
| Appearance: | Colorless to slightly yellow solid |
|---|---|
| Solubility in Dimethyl sulfoxide: | To pass test (clear, colorless to slightly yellow) |
| NMR spectrum: | Authentic |
| -20°C, Protect from light |














