Cytotoxicity LDH Assay Kit-WST

Cytotoxicity Assay
- Can be used with and without transferring supernatant
- Stable Working Solution (6 months at 0-5 oc)
- No need to freeze the Working Solution
-
Product codeCK12 Cytotoxicity LDH Assay Kit-WST
| Unit size | Price | Item Code |
|---|---|---|
| 100 tests | CK12-01 | |
| 500 tests | CK12-05 | |
| 2000 tests | CK12-20 |
| 100 tests | ・Dye Mixture ・Assay Buffer ・Lysis Buffer ・Stop Solution |
×1 11 ml×1 1.1 ml×1 5.5 ml×1 |
|---|---|---|
| 500 tests | ・Dye Mixture ・Assay Buffer ・Lysis Buffer ・Stop Solution |
×1 55 ml×1 5.5 ml×1 27.5 ml×1 |
| 2000 tests | ・Dye Mixture ・Assay Buffer ・Lysis Buffer ・Stop Solution |
×4 55 ml×4 5.5 ml×4 27.5 ml×4 |
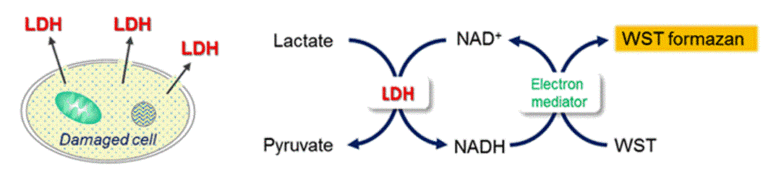
Detection Principle
Lactate dehydrogenase(LDH) is an enzyme that presents in almost cell types and it catalyzes the oxidation of lactate to pyruvate in the presence of co-enzyme NAD+. Once cells are impaired by stress, injuries, chemicals, or intercellular signals, LDH is rapidly released from the cell membrane. Thus, the measurement of the amount of released LDH from cells is one of the major methods to assess the cell death. Since Dojindo’s Cytotoxicity LDH Assay Kit-WST neither reflects the activity of living cells nor is harmful to cells, it allows the assay to perform in wells containing both viable and damaged cells.
Choose between Two Procedures
Cytotoxicity LDH Assay Kit-WST can be applied with and without supernatant transferring. Please choose suitable method for your experiment.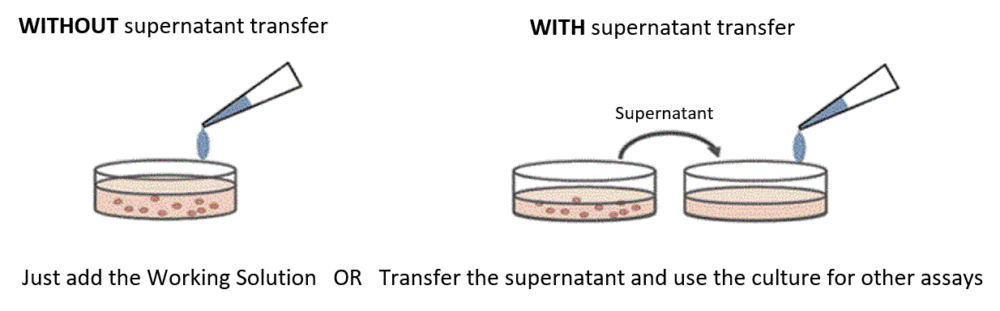
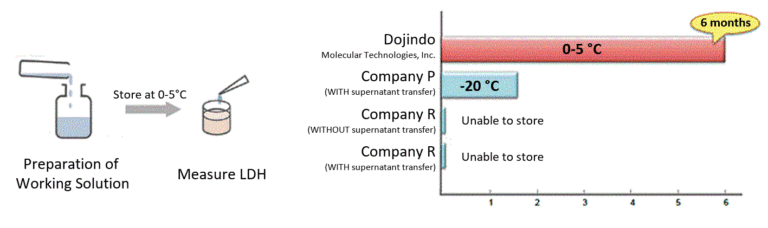
Stable Working Solution
Working Solution is stable for 6 months under refrigerated conditions. Therefore, after the preparation, Working Solution can be used as a ready-to-use solution at any time during this period.
Experimental Examples
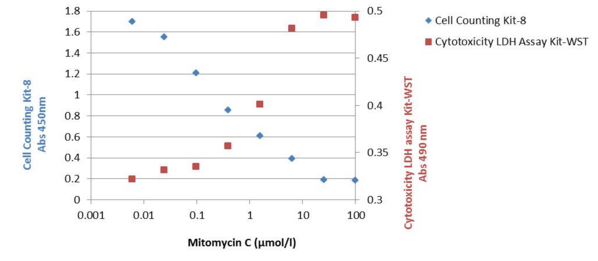
To obtain an accurate result in cytotoxicity assay, the samples are measured by different principles. Cell Counting Kit-8 (Product Code: CK04) measure NADH in living cells and LDH assay measure released LDH from dead cells. According to these results, the living cells decreased and dead cells increased with higher concentration of toxicant.
| Developer | Dojindo Molecular Technologies, Inc. |
|---|
Manual
Homogeneous Assay
Non-homogeneous Assay
Optimization of cell concentration
Technical info
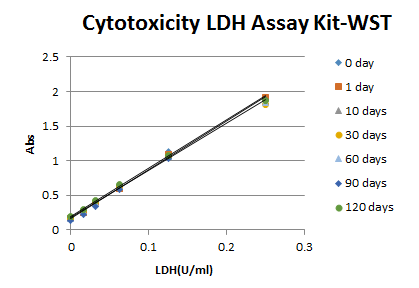
Reconstituted Working Solution has higher stability than competitor’s kit, eliminating the additional time to prepare the Working Solution for each assay.
Comparison of Working Solution Stability

Linearity
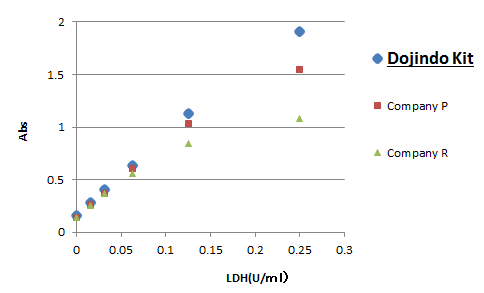
Dojindo’s Cytotoxicity LDH Assay Kit-WST has higher sensitivity than competitor’s kits
Cytotoxicity of mitomycin C using HeLa cells

Culture medium: MEM, 10% FBS
Incubation: 37C, 5% CO2, 48 hours
References
| Reference No. | Sample | Citation |
|---|---|---|
| 1) | Cell (C3H10T1/2) |
S. F. Jin, H. L. Ma, Z. L. Liu, S. T. Fu, C. P Zhang, Y. He, "XL413, a cell division cycle 7 kinase inhibitor enhanced the anti-fibrotic effect of pirfenidone on TGF-β1-stimulated C3H10T1/2 cells via Smad2/4.", Exp Cell Res., 2015, 339, (2), 289. |
| 2) | Cell (HepG2) |
S. Watanabe, C. S. Moniaga, S. Nielsen, M. Hara-Chikuma, "Aquaporin-9 facilitates membrane transport of hydrogen peroxide in mammalian cells.", Biochem Biophys Res Commun ., 2016, 471, 191. |
| 3) | Cell (HEECs, Human Esophageal Epithelial Cells) |
L. Wu, T. Oshima, J. Shan, H. Sei, T. Tomita, Y. Ohda, H. Fukui, J. Watari, H. Miwa , "PAR-2 activation enhances weak acid-induced ATP release through TRPV1 and ASIC sensitization in human esophageal epithelial cells.", Am J Physiol Gastrointest Liver Physiol ., 2015, 309, G695. |
| 4) | Cell (Human L02 Hepatocyte) |
D. Zhou, B-H Li, J. Wang, Y-N Ding, Y. Dong, Y-W Chen and J-G Fan, "Prolyl Oligopeptidase Inhibition Attenuates Steatosis in the L02 Human Liver Cell Line.", PloS ONE., 2016, 11, (10), e0165224. |
| 5) | Cell (Human primary Hepatocyte) |
T. Fukami, A. Iida, K. Konishi, M. Nakajima , "Human arylacetamide deacetylase hydrolyzes ketoconazole to trigger hepatocellular toxicity", Biochem. Pharmacol., 2016, 116, 153. |
| 6) | Cell (Mouse embryonic fibroblast:MEF) |
Y. Takanezawa, R. Nakamura, Y. Sone, S. Uraguchi and M. Kiyono, "Atg5-dependent autophagy plays a protective role against methylmercury-induced cytotoxicity", Toxicol. Lett., 2016, 262, 135. |
| 7) | Cell (neural stem/progenitor cells) |
M. Tanaka, M. Yoneyama, T. Shiba, T. Yamaguchi, K. Ogita, "Protease-activated receptor-1 negatively regulates proliferation of neural stem/progenitor cells derived from the hippocampal dentate gyrus of the adult mouse ", J Pharmacol Sci., 2016, 131, (3), 162 |
| 8) | Cell (Primary cultured cortical neurons) |
S. Wakatsuki, A. Furuno, M. Ohshima, and T. Araki, "Oxidative stress-dependent phosphorylation activates ZNRF1 to induce neuronal/axonal degeneration", J Cell Biol., 2015, 211, (4), 881. |
| 9) | Cell (DLD1 and AGS cells) |
A. Ishijima, K. Minamihata, S. Yamaguchi, S. Yamahira, R. Ichikawa, E. Kobayashi, M. Iijima, Y. Shibasaki, T. Azuma, T. Nagamune & I. Sakuma, "Selective intracellular vaporisation of antibody-conjugated phase-change nano-droplets in vitro", Scientific Reports ., 2017, DOI:10.1038/srep44077. |
| 10) | Cell (U87, HUVEC cell) |
N. Li, W. Zhang, M. Khan, L. Lin, JM. Lin, "MoS2-LA-PEI nanocomposite carrier for real-time imaging of ATP metabolism in glioma stem cells co-cultured with endothelial cells on a microfluidic system", Biosens Bioelectron., 2017, 99, (2018), 142. |
| 11) | Tissue (Mouse liver) |
Y. Sakurai, W. Mizumura, M. Murata, T. Hada, S. Yamamoto, K. Ito, K. Iwasaki, T. Katoh, Y. Goto, A. Takagi, M. Kohara, H. Suga, and H. Harashima, "Efficient siRNA delivery by lipid nanoparticles modified with a non-standard macrocyclic peptide for EpCAM-targeting", Mol. Pharm.., 2017,10.1021/acs.molpharmaceut.7b00362. |
| 12) | Cell (Human pleural mesothelial cell) |
K. Hattori, K. Nakadate, A. Morii, T. Noguchi, Y. Ogasawara, K. Ishii., "Exposure to nano-size titanium dioxide causes oxidative damages in human mesothelial cells: The crystal form rather than size of particle contributes to cytotoxicity.", Biochem. Biophys. Res. Commun.., 2017,10.1016/j.bbrc.2017.08.054. |
| 13) | Tissue (murine distal colon) |
H. Sakai, S. Tabata, M. Kimura, S. Yabe, Y. Isa, Y. Kai, F. Sato, T. Yumoto, K. Miyano, M. Narita and Y. Uezono, "Active Ingredients of Hange-shashin-to, Baicalelin and 6-Gingerol, Inhibit 5-Fluorouracil-Induced Upregulation of CXCL1 in the Colon to Attenuate Diarrhea Development", Biol. Pharm.Bull.., 2017, 40, (12), 2134. |
| 14) | Cell (SH-SY5Y cell) |
R. Watanabe, T. Kurose, Y. Morishige and K. Fujimori, "Protective Effects of Fisetin Against 6-OHDA-Induced Apoptosis by Activation of PI3K-Akt Signaling in Human Neuroblastoma SH-SY5Y Cells.", Neurochem. Res.., 2018, 43, (2), 488-499. |
| 15) | Cell (MC3T3-E1[Derived from mouse cranial crown]) |
A. Oyane, M. Nakamura, I. Sakamaki, Y. Shimizu, S. Miyata and H. Miyaji, "Laser-assisted wet coating of calcium phosphate for surface-functionalization of PEEK", PLoS ONE ., 2018, doi:10.1371/journal.pone.0206524. |
| 16) | Cell CRC Cell [Colorectal cancer]、HT29 Cell[Adenocarcinoma of the human colon] |
T. Fuji, Y. Umeda, A. Nyuya, F. Taniguchi, T. Kawai, K. Yasui, T. Toshima, K. Yoshida, T. Fujiwara, A. Goel and T.Nagasaka, "Detection of Circulating MicroRNAs with Ago2 Complexes to Monitor the Tumor Dynamics of Colorectal Cancer Patients during Chemotherapy.", Int. J. Cancer., 2018, doi: 10.1002/ijc.31960 . |
| 17) | CEll (SFXN2 knockout HEK293) |
E. E. Mon, F. Y. Wei, R. N. R. Ahmad, T. Yamamoto, T. Moroishi and K. Tomizawa, "Regulation of mitochondrial iron homeostasis by siderofexin 2 ", J Physiol Sci., 2018, doi:10.1007/s12576-018-0652-2. |
| 18) | Cell (Human umbilical vein cell line [EA.Hy926]) |
Y. Xu, C. Huang, M. Liu, N. Chen, W. Chen, C. Yang, Y. Zhao, X. Li, J. Duan, S. Liu and S. Yang, "Survivin regulated by autophagy mediates hyperglycemia-induced vascular endothelial cell dysfunction", Exp. Cell Res.., 2018, doi: 10.1016/j.yexcr.2018.01.037. |
| 19) | Cell (MIAPaCa-2[Human Pancreatic Cancer]) |
M. Akimoto, R. Maruyama, Y. Kawabata, Y. Tajima and K. Takenaga, "Antidiabetic adiponectin receptor agonist AdipoRon suppresses tumour growth of pancreatic cancer by inducing RIPK1/ERKdependent necroptosis", Cell Death Dis., 2018, 9, 804. |
| 20) | Cell (Fibro adipogenic progenitor cells:FAPs) |
Y. Saito, T. Chikenji, T. Matsumura, M. Nakano and M. Fujimiya, "Exercise enhances skeletal muscle regeneration by promoting senescence in fibro-adipogenic progenitors", Nat. Commun., 2020, doi:10.1038/s41467-020-14734-x. |
Q & A
-
Q
Can I measure with filters other than 490 nm?
-
A
We recommend 490 nm, but a difference of 490±2-3 nm is no problem. 450 nm filters can be used as we have experience with them. Please note that the absorbance will be higher than with the 490 nm filter.
-
Q
Is there any correlation with Cell Counting Kit-8 in toxicity testing?
-
A
Cytotoxicity LDH Assay Kit-WST and Cell Counting Kit-8 do not necessarily correlate with each other due to their different measurement principles. Therefore, when testing for cytotoxicity, it is recommended to use several types of indicators, such as the LDH Assay and the Cell Counting Kit-8.
- Cytotoxicity LDH Assay Kit-WST: Measures LDH activity released into the medium due to cell membrane damage (cell membrane damage is an indicator).
- Cell Counting Kit-8: Measures intracellular metabolic activity (dehydrogenase activity is an indicator).
-
Q
The LD50 value differs significantly from previous measurements. Is there anything I can do?
-
A
The LD50 value may be affected by the test substance depending on the state of the cells. The LD50 value is affected by the state of the cells.
The LD50 value may be more reliable if a narrower dilution factor is used when preparing a dilution series of the test substance.
It is recommended that the concentration of the test substance to be used be determined according to the following procedure.<Study procedure>
(1) Repeat the 10-fold dilution based on the highest available concentration of the test substance and perform toxicity testing over a wide sample concentration range of about 5-6 digits. [Figure 1]. Using the operation in (1), make a rough estimate of the highest predicted concentration in the concentration range at which toxicity will occur.
(2) Based on the result of (1), repeat 2-fold dilution based on the predicted maximum concentration (the lowest concentration that causes 100% lethality) at which the concentration of the substance to be tested appears, and perform toxicity testing in a concentration range covering 2-3 orders of magnitude. [Figure 2].
(3) Repeat step (2) to finally establish a concentration range in which at least 3 sample concentrations between 20-80% cytotoxicity can be plotted. [Figure 3]
-
Q
Can you measure different isoenzymes of LDH?
-
A
This product is designed to measure cytotoxicity using total LDH activity as an indicator, so it cannot measure LDH1, LDH2, LDH3, etc. separately.
-
Q
Can I store samples?
-
A
It can be stored for approximately one week in refrigerated storage1).
Storage at room temperature2) or freezing is not recommended due to inactivation of LDH.
1) M. Allen, P. Millett, E. Dawes, N. Rushton., "Lactate dehydrogenase activity as a rapid and sensitive test for the quantification of cell numbers in vitro.", Clin Mater. 1994; 16, (4), 189.
2) A. Wagner, A. Marc, JM. Engasser, A. Einsele, "The use of lactate dehydrogenase (LDH) release kinetics for the evaluation of death and growth of Biotechnol Bioeng. 1992; 39, (3), :320.Stability may vary depending on evaluation conditions and other factors. Please refer to the above paper with understanding.
-
Q
Does the Cytotoxicity LDH Assay Kit-WST include LDH standards?
-
A
No, LDH standards are not included in the kit as the kit is intended for cytotoxicity testing.
-
Q
Is it necessary to test immediately after adding Stop Solution?
-
A
Yes, the assay should be performed within 3 hours after the addition of the Stop Solution. In this case, please protect the plate from light.
-
Q
There is a lot of variation between wells, what could be the cause?
-
A
There is a lot of variation between wells, what could be the cause?
Please check the following possible reasons.
(1) Air bubbles are formed on the surface of the culture medium.
Please remove the air bubbles using the following methods.
Remove air bubbles with the tip of a syringe or syringe needle.
(If you have a 96-well plate centrifuge) Centrifuge at 1,000 x g for 1 minute.(2) The reagent concentration has changed due to evaporation of water from the culture medium during incubation.
The outermost wells of the microplate are prone to water evaporation during incubation. When incubating for a long time, do not use the outermost wells for measurement, but use only the culture medium in them.
(3) When adding reagents, there is a large time difference between the first and last wells to be added.
For Working Solution and Stop Solution, which are added to all wells, the use of a multichannel pipette is recommended.
(4)The added reagents are not well mixed.
When adding Lysis Buffer, Working Solution and Stop Solution, mix them with a plate mixer or gently tap the side of the plate with your finger to mix the added reagents and medium.
Note that the amount of Lysis Buffer added is small and will affect the High Control value.
When tapping the plate, be careful not to spill the medium in the wells.(5) The weight of the (multichannel) pipette used is not accurate.
Please check if the pipette weight is accurate.
-
Q
The background is now high. What is the cause and what can I do about it?
-
A
The following causes are possible.
(1) LDH present in serum in the medium is increasing the background. Use serum-free medium or medium prepared with 5% or less serum.
(2) Strong reducing substances in the test substance or medium may cause the WST dye to miscolorize and increase the background. Confirm this by measuring a reagent blank.
(In addition, common culture media such as DMEM, RPMI, F-12, etc. usually do not contain reducing agents).
-
Q
Small difference in absorbance between high and low controls during cell count optimization.
-
A
(1) The absorbance of the low control may be higher due to high background. Check the following
LDH in serum in the medium increases the background. Use serum-free medium or medium prepared with 5% or less serum.
If the test sample or culture medium contains strong reducing substances, the WST dye will mischromatize and increase the background. Confirm this by measuring a reagent blank.(2) The High Control may contain viable cells.
To ensure that the Lysis Buffer is evenly distributed in the wells and that all cells are dead cells, gently vortex the plate after adding the Lysis Buffer to mix the Lysis Buffer thoroughly.
Handling and storage condition
| 0-5°C, Protect from light | |
|
Danger / harmful symbol mark |
 
|
|---|---|




















