Cellular Senescence Plate Assay Kit - SPiDER-βGal

Senescence Cell Detection (for Microplate)
-
Product codeSG05 Cellular Senescence Plate Assay Kit - SPiDER-βGal
| Unit size | Price | Item Code |
|---|---|---|
| 20 tests | SG05-01 | |
| 100 tests | SG05-05 |
| 20 tests | SPiDER-βGal Lysis Buffer Assay Buffer Stop Solution |
×1 40 ml×1 1.5 ml×1 3 ml×1 |
|---|---|---|
| 100 tests | SPiDER-βGal Lysis Buffer Assay Buffer Stop Solution |
×5 100 ml×2 7.5 ml×1 15 ml×1 |
Description
Description:
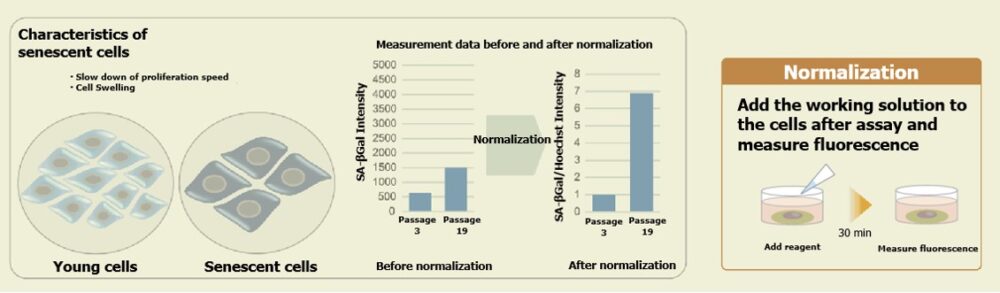
This product is a simple detection kit by plate assay for senescence-associated β-galactosidase (SA-β-gal) activity which is used as a marker for senescent cells. By simply adding SPiDER-βGal, a reagent for detection of β-galactosidase, to 96 well plates, this kit allows you to quantify SA-β-gal activity and makes it possible to evaluate multiple specimens. When normalization is done by the results obtained by counting cells, quantifying nucleic acids (the relevant product), or quantifying proteins, the measured values obtained using this kit become available for evaluating SA-β-gal activity according to cell number.
Technical info
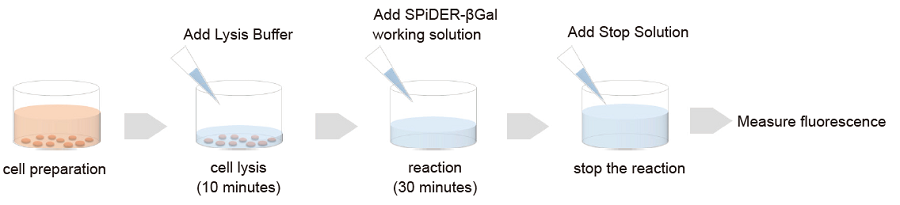
Cells prepared in advance are lysed in the buffer supplied with this kit.Fluorescence intensity is obtainable according to SA-β-gal activity simply by adding the fluorescent substrate SPiDER-βGal to the cell lysate. Even when you prepare cells in 100 mm dishes or others, fluorescence intensity can be measured by transferring cell lysate in 96 well plates after cell lysis.
Correlation with imaging data:
Imaging assessments of WI-38 cells at different passage levels were performed with this Plate Assay Kit and the Cellular Senescence Detection Kit – SPiDER-βGal
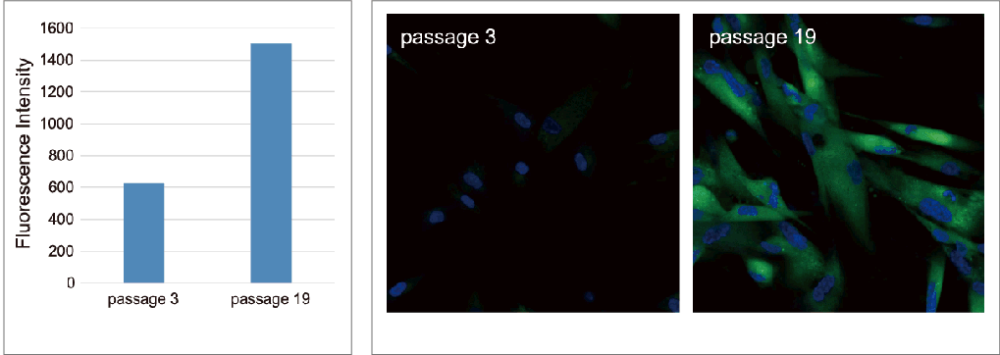
As a result, it was confirmed that in both kits, SA-β-gal staining increased in the high-passage WI-38 cells.
Bear in mind that although initial cell seeding densities are the same, cell densities at the time of plate assay differ due to low proliferation rate of senescent cells at higher passage levels. Therefore, in this experiment, we used SA-β-Gal activity values normalized by the results obtained using the Cell Count Normalization Kit (coming soon) in which cell number is determined by a nuclear marker.
Plate Assay
Ex. 535nm / Em. 580nm
Imaging data
Green: Ex. 488nm / Em. 500-600nm (SA-β-Gal staining with Cellular Senescence Detection Kit – SPiDER-βGal)
Blue: Ex. 405nm / Em. 450-495nm (Nuclear staining with -Cellstain- DAPI solution)
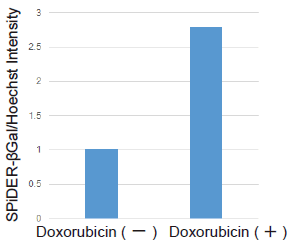
Evaluation doxorubicin-treated with cells
Cell counts may need to be normalized. When cells are analyzed in a microplate, the results obtained may sometimes differ depending on cell numbers per well.In such cases, normalization of the measured values obtained from cell counting and total protein will be necessary. In this kit, cell numbers can be easily measured by the fluorescence intensity induced by a reagent added to cell culture medium for staining nuclei.
Ex. 535nm / Em. 580nm
Precautions when using this kit
Cell counts may need to be normalized. When cells are analyzed in a microplate, the results obtained may sometimes differ depending on cell numbers per well. In such cases, normalization of the measured values obtained from cell counting and total protein will be necessary. In the Cell Count Normalization Kit, cell numbers can be easily measured by the fluorescence intensity induced by a reagent added to the cell culture medium for staining nuclei.
References
| No. | Sample | Instrument | Reference(Link) |
|---|---|---|---|
| 1 | Cell (MRC-5) |
Plate Reader | Y. Takenaka, I. Inoue, T. Nakano, M. Ikeda and Y. Kakinuma, "Prolonged disturbance of proteostasis induces cellular senescence via temporal mitochondrial dysfunction and subsequent mitochondrial accumulation in human fibroblasts", 2021, doi:10.1111/febs.16249. |
| 2 | Cell (hRPE) |
Plate Reader | H. Yamamoto-Imoto, S. Minami, T. Shioda, Y. Yamashita, S. Sakai, S. Maeda, T. Yamamoto, S. Oki, M. Takashima, T. Yamamuro, K. Yanagawa, R. Edahiro, M. Iwatani, M. So, A. Tokumura, T. Abe, R. Imamura, N. Nonomura, Y. Okada, D. E. Ayer, H. Ogawa, E. Hara, Y. Takabatake, Y. Isaka, S. Nakamura and T. Yoshimori, "Age-associated decline of MondoA drives cellular senescence through impaired autophagy and mitochondrial homeostasis", Cell Rep., 2022, doi:10.1016/j.celrep.2022.110444. |
| 3 | Cell (U251MG) |
Plate Reader | A. Saito, Y. Kamikawa, T. Ito, K. Matsuhisa, M. Kaneko T. Okamoto, T. Yoshimaru, Y. Matsushita, T. Katagiri and K. Imizumi, "p53-independent tumor suppression by cell-cycle arrest via CREB/ATF transcription factor OASIS", 2023, doi:10.1016/j.celrep.2023.112479. |
Q & A
-
Q
How many samples can be measured with this kit?
-
A
・20 tests: 6 samples (n=3)
・100 tests: 30 samples (n=3)
-
Q
Is it possible to induce cell senescence on a 96 well plate and measure it?
-
A
It is recommended to induce senescence in a 100 mm dish, etc., and then transfer the cells to a 96-well plate for measurement.
The reason is the following:
1. If senescence induction is performed in 96 well plates, the control cells may proliferate too much (depending on the number of days of senescence induction), leading to confluency.
2. Also, if a low number of cells are seeded in advance to avoid confluency, senescent cells may not have the number of cells required for the plate assay.
Handling and storage condition
| 0-5°C, Protect from light | |
|
Danger / harmful symbol mark |
   
|
|---|---|