Glucose Assay Kit-WST

Glucose Metabolism Assay
-
Product codeG264 Glucose Assay Kit-WST
| Unit size | Price | Item Code |
|---|---|---|
| 50 tests | G264-05 | |
| 200 tests | G264-20 |
| 50 tests | ・Dye Mixture ・Glucose Standard ・Enzyme ・Assay Buffer ・Reconstitution Buffer |
×1 150 μl×1 ×1 3.5 ml×1 350 μl×1 |
|---|---|---|
| 200 tests | ・Dye Mixture ・Glucose Standard ・Enzyme ・Assay Buffer ・Reconstitution Buffer |
×1 600 μl×1 ×1 14 ml×1 1.4 ml×1 |
Description
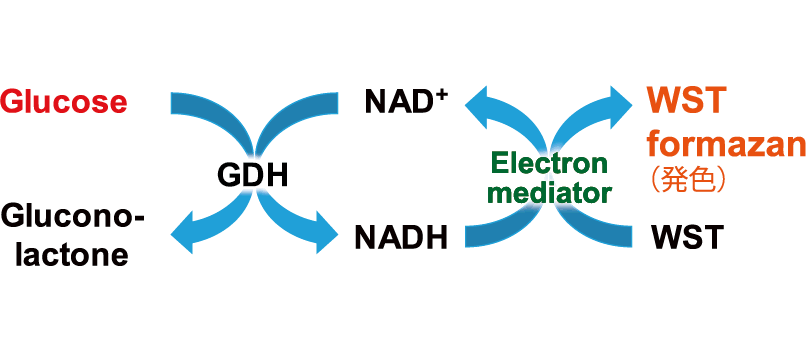
Principle
This kit can detect glucose found within cell culture medium or intracellular glucose by measuring the absorbance of the colored WST formazan dye. The degree of absorbance depends on the amount of glucose present within the sample.This kit contains a glucose standard solution that can be used to create a standard curve. This allows for the quantitation of glucose levels present in the sample.
Manual
Technical info
Procedure
Procedures are so easy that you simply incubate the plate after the addition of culture supernatant or tissue/cell lysates prior to adding a reagent.

Supplementary information on technology and products used
Preparation of Standard Curve
Glucose levels in a sample can be measured by a calibration curve established with Glucose Standards included in this kit. If the glucose levels are greater than or equal to 0.5 mmol/L, the sample must be diluted before measurement.
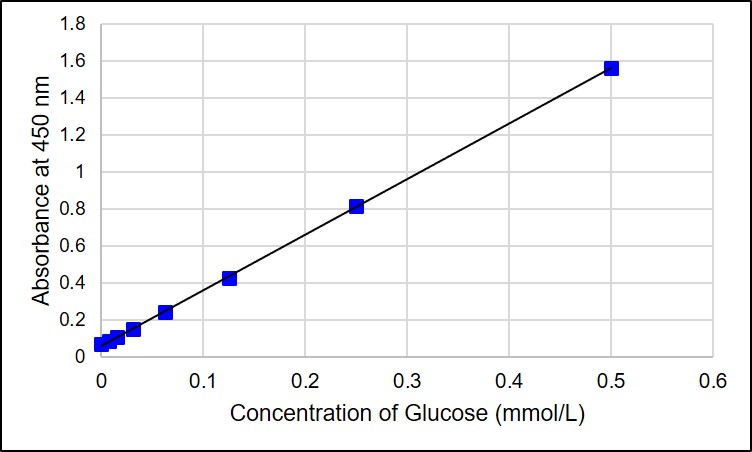
Supplementary information on technology and products used
Measurement of Glucose level in combination with Lactate Assay Kit
By using Glucose Assay Kit-WST and Lactate Assay Kit-WST, we have successfully measured metabolic activity changes of phloretin, a protein transport inhibitor, when it is added to Jurkat cells.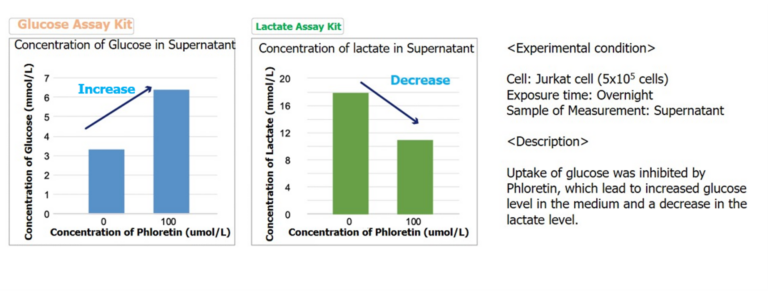
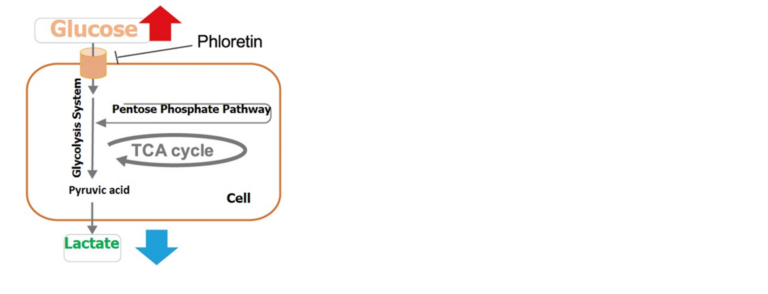
Precautions when using this kit.
Cell counts may need to be normalized. When cells are analyzed in a microplate, the results obtained may sometimes differ depending on cell numbers per well. In such cases, normalization of the measured values obtained from cell counting and total protein will be necessary. In our Cell Count Normalization Kit, cell numbers can be easily measured by the fluorescence intensity induced by a reagent added to the cell culture medium for staining nuclei.
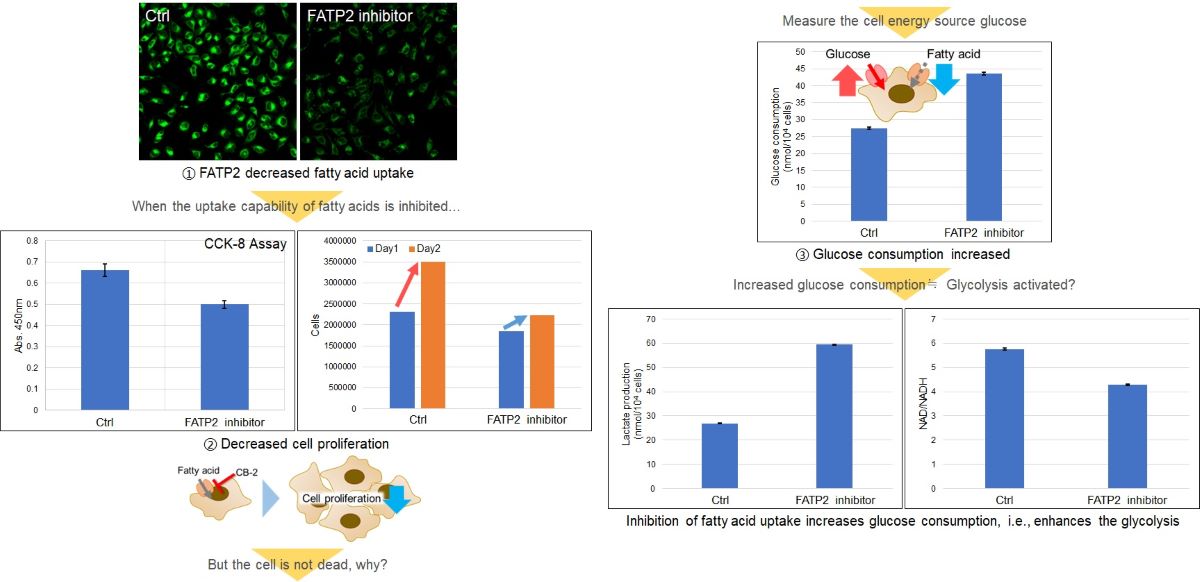
Application Data: Alteration of intracellular metabolism of HeLa cells by treatment with fatty acid transporter inhibitors
Fatty acids are important for membrane synthesis and are essential for cell proliferation. HeLa cells were treated with a fatty acid transporter inhibitor, and then the cell proliferation and intracellular metabolism (glucose consumption, Lactate release, and NAD/NADH ratio) were tested.
The results showed a decrease in cell proliferation, an increase in glucose consumption and Lactate release, and a decrease in the intracellular NAD+/NADH ratio, indicating a shift in the metabolic pathway to glycolysis.
<Related Products>
Free Acid Uptake: Fatty Acid Uptake Assay Kit (Code: UP07)
Cell Proliferation: Cell Counting Kit-8 (Code: CK04)
Lactate: Lactate Assay Kit-WST (Code: L256)
NAD/NADH Ratio: NAD/NADH Assay Kit-WST (Code: N509)
References
| Reference No. | Sample | Cite |
|---|---|---|
| 1) | Serum (Mouse) |
M. Shinohara, Y. Tashiro, M. Shinohara, J. Hirokawa, K. Suzuki, M. O. Takeya, M. Mukouzono, S. Takeda, T. Saito, A. Fukumori, T. C. Saido, R. Morishita and N. Sato, "Increased levels of Aβ42 decrease the lifespan of ob/ob mice with dysregulation of microglia and astrocytes”, FASEB J., 2019,DOI: 10.1096/fj.201901028RR |
| 2) | Microorganism (Streptomyces albulus ) |
K. Yamanaka, Y. Hamano and T. Oikawa, "Enhancement of metabolic flux toward ε-poly-l-lysine biosynthesis by targeted inactivation of concomitant polyene macrolide biosynthesis in Streptomyces albulus.”, J. Biosci. Bioeng., 2020,DOI: 10.1016/j.jbiosc.2019.12.002 |
| 3) | Cell (HCT116) |
K. Ohshima, S. Nojima, S. Tahara, M. Kurashige, K. Kawasaki, Y. Hori, M. Taniguchi, Y. Umakoshi, D. Okuzaki, N. Wada, J. Ikeda, E. Fukusaki and E. Morii, "Serine racemase enhances growth of colorectal cancer by producing pyruvate from serine”, Nat Metab, 2020, 2(1), 81 |
| 4) | Cell (P388 Leukemia) |
T. Matsuo, Y. Konya, E. Hirayama and Y. Sadzuka , "2-Deoxy-D-glucose enhances the anti-cancer effects of idarubicin on idarubicin-resistant P388 leukemia cells”, Oncol Lett , 2020, 20(1), 962-966 |
| 5) |
Cells (mouse: sperm) |
M. Hashimoto, S. Kimura, C. Kanno, Y. Yanagawa, T. Watanabe, J. Okabe, E. Takahashi, M. Nagano and H. Kitamura, "Macrophage ubiquitin‑specific protease 2 contributes to motility, hyperactivation, capacitation, and in vitro fertilization activity of mouse sperm”, Cellular and Molecular Life Sciences, 2020, doi: 10.1007/s00018-020-03683-9 |
| 6) | Cells (Macrophages) |
N. Saeki and Y. Imai, "Reprogramming of synovial macrophage metabolism by synovial fbroblasts under infammatory conditions ”, Cell Commun Signal, 2020, 18, 188 |
Q & A
-
Q
How many samples can I measure?
-
A

* The number of samples that can be recorded when the standard curve and sample is measured in triplicates.
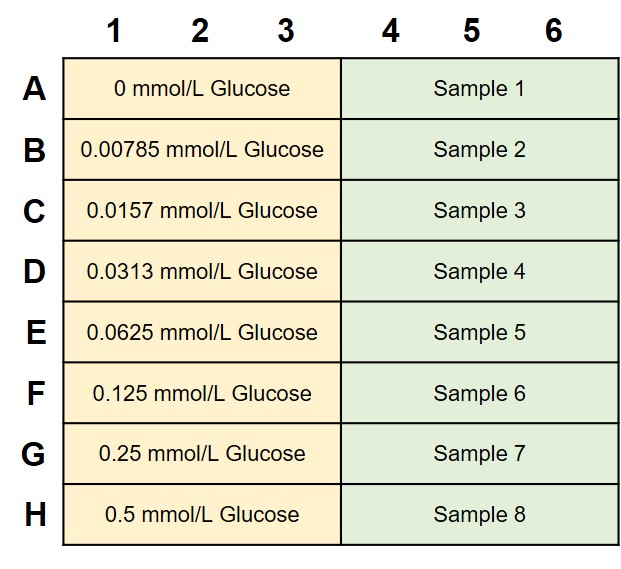
*A plate layout for Glucose standard solution and sample
-
Q
I do not have a 450 nm filter. What other filters can I use?
-
A
You can use filters with an absorbance between 450 nm and 490 nm. However, the absorbance value can become lower if the sample is not measured at 450 nm.
-
Q
Can I measure L-Glucose levels with this Kit?
-
A
No, this kit is used for the measurement of β-D-Glucose levels.
-
Q
Does this kit detect 2-deoxy-D-glucose, a glucose analog?
-
A
Yes. 2-deoxy-D-glucose is also detected as a glucose.
-
Q
Can samples other than cell culture supernatant be measured?
-
A
Intracellular glucose levels can be measured with this kit. Please refer to Q&A "Can I measure intracellular glucose levels with this kit?". Unfortunately, there are no examples of other samples.
-
Q
Can I measure intracellular glucose levels with this kit?
-
A
Yes, you can measure intracellular glucose with this kit.
You need to prepare "0.1% Triton X-100 solution" and "ultrafiltration filter (MWCO:10K)" for the measurement.
Please follow the following protocol.(1) Prepare the cells*1 in a 1.5 ml microtube.
*The proper number of cells required for the assay depends on the cell line. For HepG2 and Jurkat cells please refer to the following figure.(2) Centrifuge at 300 x g for 5 min. Remove the supernatant. (3) Add 300 µl cold PBS, after pipetting, and centrifuge at 300 x g for 5 min. Remove the supernatant. (4) Add 250 µl of cell lysate*2 (0.1% Triton X-100 solution), dissolve the cells by pipetting, and then centrifuge at 12,000 x g for 5 min. (5) Transfer 200 µl of the supernatant from step (4) to an ultrafiltration membrane filter (MWCO: 10K) and centrifuge at 12,000x g for 10 min.
*At least 150 µl total sample is required for n=3 (50 µl per well x 3 wells).
*If the filtrate is under 150 µl, extend the centrifugation time.(6) Take the filtrate from step (5) as a test sample.
Follow the manual to measure the glucose.
* About the test sample, please dilute the sample to the range of the standard curve(0-0.5 mmol/l) and then measure with the kit.*1To detect glucose above 0.02 mmol/l, a minimum of 1 × 105 cells for HepG2 cells and 2 × 106 cells for Jurkat cells are required.
*2SDS will affect the measurement, please use an SDS-free solution for cell lysate.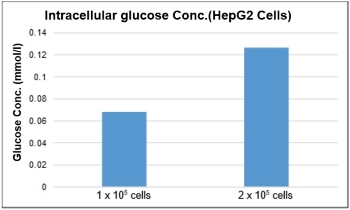
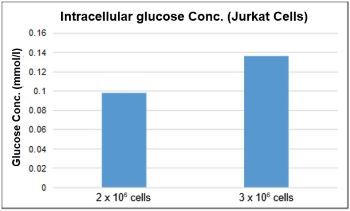
-
Q
Can I measure samples that contain a reducing agent?
-
A
You can’t measure an accurate glucose level because WST dye interferes with the reducing agent. If you add test material which possesses reducing properties, please prepare a few wells that only contain the medium without cells and only test material for background control.
-
Q
How long is the working solution stable?
-
A
The working solution protected from light is stable at room temperature for about 4 hours. Please prepare it immediately prior to use. The working solution changes its color from red to orange when it is exposed to light. High background absorbance may result if the orange-colored working solution is used.
-
Q
How long can samples be stored?
-
A
We have confirmed that cell culture supernatant can be stored for 3 weeks at -20 degrees, and cell lysate for 1 month at -20 degrees. As for the cell lysate, it is necessary to removal of proteins from the samples prior to store. Please refer to Q&A: "Can I measure intracellular glucose levels with this kit?" step (5).
-
Q
Our samples did not change in color, are there any reasons for this?
-
A
Glucose level contained in the sample may be lower than the detection limit (0.02 mmol/l) that can be measured using this kit. If you dilute the sample, the glucose level contained in the diluted sample may be lower than 0.02 mmol/l. In this case, please lower the dilution ratio and adjust the concentration of the sample over the detection limit that was measured using this kit.
Handling and storage condition
| 0-5°C, Protect from light, Protect from moisture |















