Lactate Assay Kit-WST

Lactic Acid Measurement
-
Product codeL256 Lactate Assay Kit-WST
| Unit size | Price | Item Code |
|---|---|---|
| 50 tests | L256-05 | |
| 200 tests | L256-20 |
| 50 tests | ・Dye Mixture ・Lactate Standard ・Enzyme Solution ・Assay Buffer ・Reconstitution Buffer |
x1 150 μl x1 12 μl x1 5.5 ml x1 550 μl x1 |
|---|---|---|
| 200 tests | ・Dye Mixture ・Lactate Standard ・Enzyme Solution ・Assay Buffer ・Reconstitution Buffer |
x1 600 μl x1 48 μl x1 11 ml x2 2.2 ml x1 |
Description
Lactate Assay Kit-WST can quantify lactate (the lower limit: 0.02 mmol/l), one of the glycolysis metabolites. Because the Lactate Assay Kit-WST corresponds to 96 well micro-plates, it allows for multiple samples to be measured at once. Lactate quantification has been widely used as a marker for monitoring changes in intracellular metabolic pathway in many different areas of cancer research. In recent years, there have been an increasing number of reports on using lactate as an indicator of stem cell differentiation or as an indicator in diabetic research.
Manual
Technical info
The kit is used to detect lactate in cell culture medium by measuring the absorbance of WST formazan produced according to quantities of lactate. The kit includes a “Lactate Standard” which can be used to quantify the concentration of lactate found within samples by creating standard curve.

Simple Assay Procedure

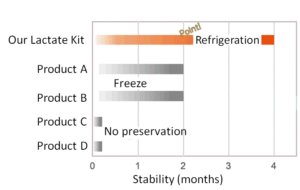
Stability
Dojindo’s stock solution is stable for 4 months under refrigerator. Your assay can be done anytime without the need freeze the product and wait for defrosting.
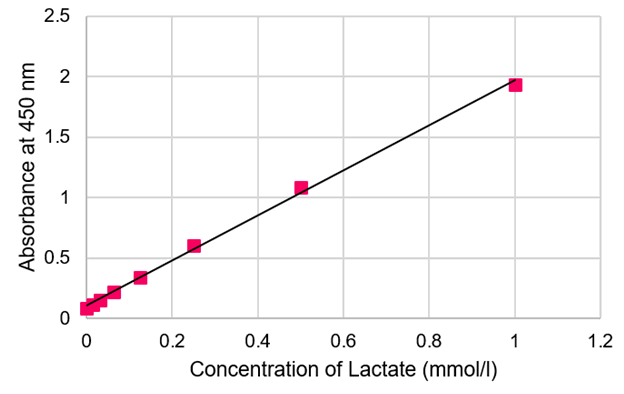
Standard curve
Standard curve can be prepared by using Lactate Standard included in the kit. The concentration of lactate can be measured. The concentration of lactate can be evaluated by diluting the samples if the concentration is over 1 mmol/l.
Measurement of Lactate level in combination with Glucose Assay Kit
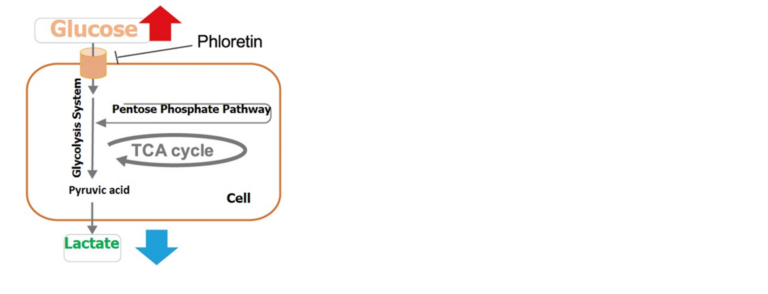
By using Glucose Assay Kit-WST and Lactate Assay Kit-WST, we have successfully measured metabolic activity changes of phloretin, a protein transport inhibitor, when it is added to Jurkat cells.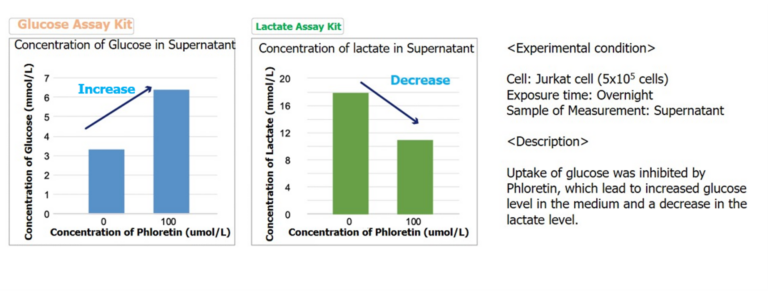
Precautions when using this kit.
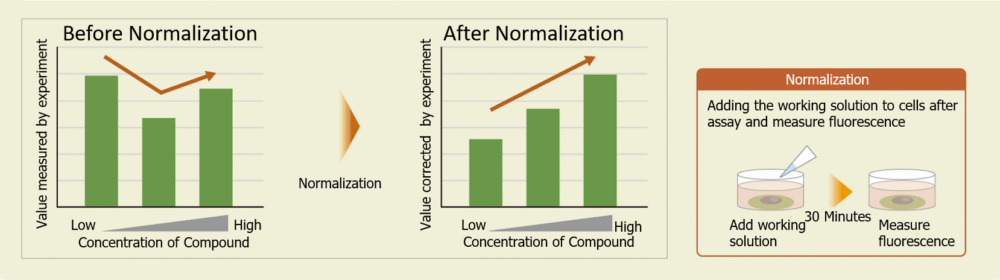
Cell counts may need to be normalized. When cells are analyzed in a microplate, the results obtained may sometimes differ depending on cell numbers per well. In such cases, normalization of the measured values obtained from cell counting and total protein will be necessary. In our Cell Count Normalization Kit, cell numbers can be easily measured by the fluorescence intensity induced by a reagent added to the cell culture medium for staining nuclei.
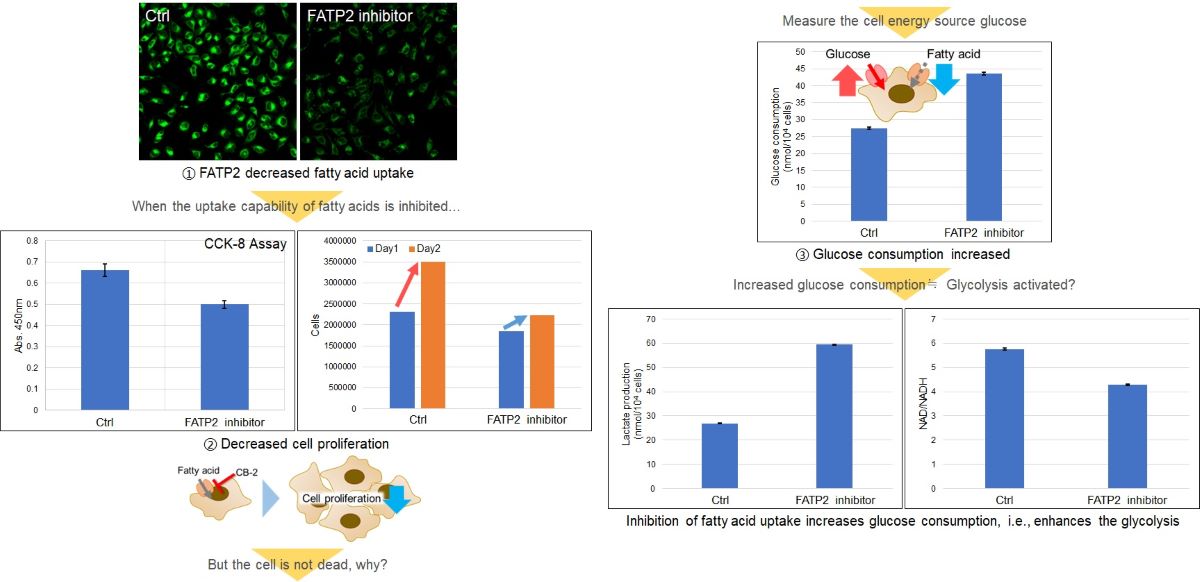
Application Data: Alteration of intracellular metabolism of HeLa cells by treatment with fatty acid transporter inhibitors
Fatty acids are important for membrane synthesis and are essential for cell proliferation. HeLa cells were treated with a fatty acid transporter inhibitor, and then the cell proliferation and intracellular metabolism (glucose consumption, Lactate release, and NAD/NADH ratio) were tested.
The results showed a decrease in cell proliferation, an increase in glucose consumption and Lactate release, and a decrease in the intracellular NAD+/NADH ratio, indicating a shift in the metabolic pathway to glycolysis.
<Related Products>
Free Acid Uptake: Fatty Acid Uptake Assay Kit (Code: UP07)
Cell Proliferation: Cell Counting Kit-8 (Code: CK04)
Glucose: Glucose Assay Kit-WST (Code: G264)
NAD/NADH Ratio: NAD/NADH Assay Kit-WST (Code: N509)
References
| No. | Sample | Publication |
|---|---|---|
| 1) | C2C12 | S. Hori, Y. Hiramuki, D. Nishimura, F. Sato and A. S. Fujisawa, PDH-mediated metabolic flow is critical for skeletal muscle stem cell differentiation and myotube formation during regeneration in mice.", FASEB J. ., 2019, 33(7), 8094. |
| 2) | A2780, OVC |
Y. Nonomiya, K. Noguchi, K. Katayama and Y. Sugimoto, Novel pharmacological effects of poly (ADP-ribose) polymerase inhibitor rucaparib on the lactate dehydrogenase pathway.", Biochem. Biophys. Res. Commun., 2019, 510(4), 510. |
| 3) | HCT116 |
K. Ohshima, S. Nojima, S. Tahara, M. Kurashige, K. Kawasaki, Y. Hori, M. Taniguchi, Y. Umakoshi, D. Okuzaki, N. Wada, J. Ikeda, E. Fukusaki and E. Morii, Serine racemase enhances growth of colorectal cancer by producing pyruvate from serine", Nat Metab, 2020, 2(1), 81. |
| 4) | Blood cell, Lymphocytes |
Y. akashima, A. Hayano and B. Yamanaka, Metabolome analysis reveals excessive glycolysis via PI3K/AKT/mTOR and RAS/MAPK signaling in methotrexate-resistant primary CNS lymphoma-derived cells.", Cancer Sci.., 2020, 111(4), 1241-1253. |
| 5) | Lung cancer |
H. Osumi, H. Horiguchi, T. Kadomatsu, K. Tashiro, J. Morinaga, T. Takahashi, K. Ikeda, T. Ito, M. Suzuki, M. Endo and Y. Oike, "Tumor Cell-Derived Angiopoietin-Like Protein 2 Establishes a Preference for Glycolytic Metabolism in Lung Cancer Cells”, Nat Metab, 2020, 2(1), 81 |
| 6) | P388 |
T. Matsuo, Y. Konya, E. Hirayama and Y. Sadzuka , "2-Deoxy-D-glucose enhances the anti-cancer effects of idarubicin on idarubicin-resistant P388 leukemia cells”, Oncol Lett , 2020, 20(1), 962-966 |
Q & A
-
Q
Can I quantify D-Lactate levels?
-
A
No, the kit is used for the quantification of L-Lactate levels.
-
Q
How stable is the Working solution?
-
A
You can’t store the Working solution. Please prepare the Working solution prior to use. Please protect from light because the Working solution is light-sensitive. The Working solution is stable for 4 hours at room temperature with protection from light.
Once the Working solution is exposed to light, the color of the solution becomes from red to orange. It caused to increase background.
-
Q
Can I measure samples that contain a reducing agent?
-
A
You can’t measure an accurate Lactate level because WST dye interferes with the reducing agent. If you add test material which possesses reducing properties, please prepare a few wells that only contain the medium without cells and only test material for background control. Please subtract the background from the absorbance of standard curve or samples
-
Q
Are there protocols for measuring intracellular lactate level?
-
A
(1) Collect cells (1 × 105 cells * 1) in a microtube.
(2) Centrifuge at 300 × g for 2 minutes and remove supernatant.
(3) Add 300 μl of cold PBS and suspend by pipetting. Centrifuge at 300 × g for 2 minutes and remove supernatant.
(4) Add 300 μl of 0.1% Triton-X * 2 and vortex for 1 minutes to prepare for cell lysate.
(5) Centrifuge at 8000 × g for 5 minutes and collect supernatant.
(6) Deproteinize the solution prepared at step (5) with an ultrafiltration membrane filter (fraction molecular weight: 10 K) * 3 to obtain a measurement sample.* 1 For HeLa cells, more than 1 × 105 cells are necessary to detect lactate level over 0.02 mmol / l.
* 2 When SDS is included in the cell Lysate, color development can be inhibited. Please avoid using buffer containing SDS.
* 3 Since endogenous Lactate dehydrogenase (LDH) causes background please remove LDH by deproteinization treatment.
-
Q
How many samples can I measure?
-
A
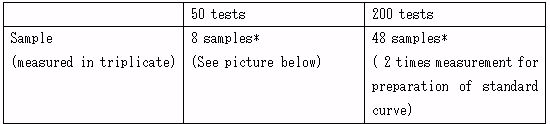
* The number of samples that can be recorded when the standard curve and sample is measured in triplicates.
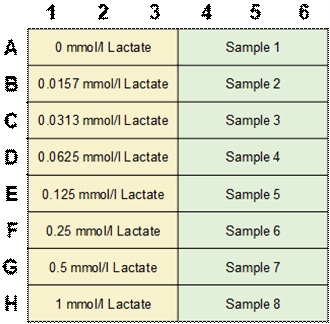
*A plate layout for Lactate standard solution and sample
-
Q
I do not have a 450 nm filter. What other filters can I use?
-
A
You can use filters with an absorbance between 450 nm and 490 nm. However, the absorbance value can become lower if the sample is not measured at 450 nm.
-
Q
Our samples did not change in color, are there any reasons for this?
-
A
Lactate level contained in the sample may be lower than the detection limit (0.02mmol/l) that can be measured using this kit. If the lactate level is below 0.02mmol/l, please consider other measurement method such as LC-MS.
If you dilute the sample, lactate level contained in the diluted sample may be lower than 0.02mmol/l.
Please lower the dilution ratio and adjust the concentration of the sample over the detection limit that was measured using this kit.
-
Q
Is it possible to measure lactate in the culture supernatant using a medium containing serum?
-
A
It is possible to measure lactate in the culture supernatant using a medium containing serum.
However, the background increases when serum is included. Therefore, only the culture medium should be measured and subtracted as a background control.
-
Q
How long can samples be stored?
-
A
We have confirmed that cell culture supernatants and cell lysates can be stored at -20°C for one month.
However, cell lysates should be stored after protein removal, e.g. by filtration.
Handling and storage condition
| 0-5°C, Protect from light |