NAD/NADH Assay Kit-WST

NAD/NADH Assay
- Easy extraction of cell Lysate
- Used to determine intracellular levels of NAD/NADH
-
Product codeN509 NAD/NADH Assay Kit-WST
| Unit size | Price | Item Code |
|---|---|---|
| 100 tests | N509-10 |
| 100 tests | ・NAD/NADH Extraction Buffer ・NAD/NADH Control Buffer ・Standard Buffer ・Assay Buffer ・Dye Mixture ・Enzyme ・Standard ・Filtration Tube |
20 ml×1 20 ml×1 10 ml×1 5.5 ml×1 550 μl×1 x1 x1 x12 |
|---|
Description
Cell lysate from a cell culture can be easily prepared via Deproteinzation using the extraction buffer and filtration tubes found within this kit. Intracellular NADH levels can be quantified by heat treatment of cell lysate. Additionally, intracellular NAD+ levels can be determined by subtracting NADH levels from independently measured total NAD+/NADH levels.
Principle
NAD/NADH Assay Kit-WST enables quantification of the amount of total NAD+/NADH, NADH and NAD+ in cells and measurement of their ratio.
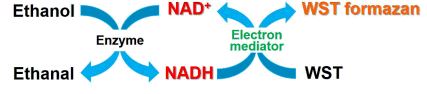
Measurement of NAD+ and NADH
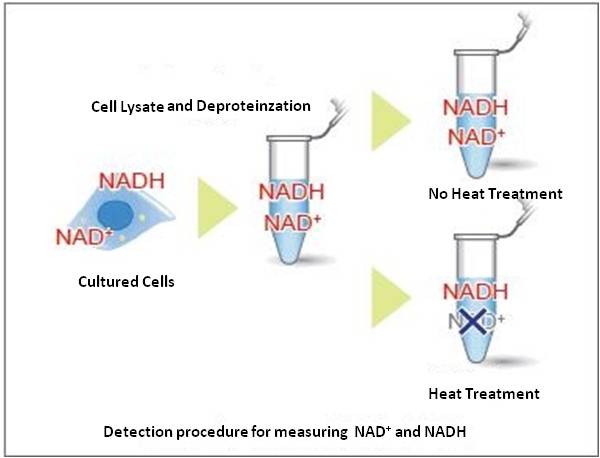
Cell lysate from a cell culture can be easily prepared via Deproteinzation using the extraction buffer and filtration tubes found within this kit. Intracellular NADH levels can be quantified by heat treatment of cell lysate. Additionally, intracellular NAD+levels can be determined by subtracting NADH levels from independently measured total NAD+/NADH levels.
Manual
Technical info
Recently, it has become clear that Sirtuin is linked to longevity and plays a role in NAD+ level regulation. Also Sirtuin has been recognized as a marker necessary to understanding biological states, such as obesity & diabetes, as well as cellular differentiation.
In the experiment shown below, the NAD+ /NADH levels & ratio were determined using HeLa cells.
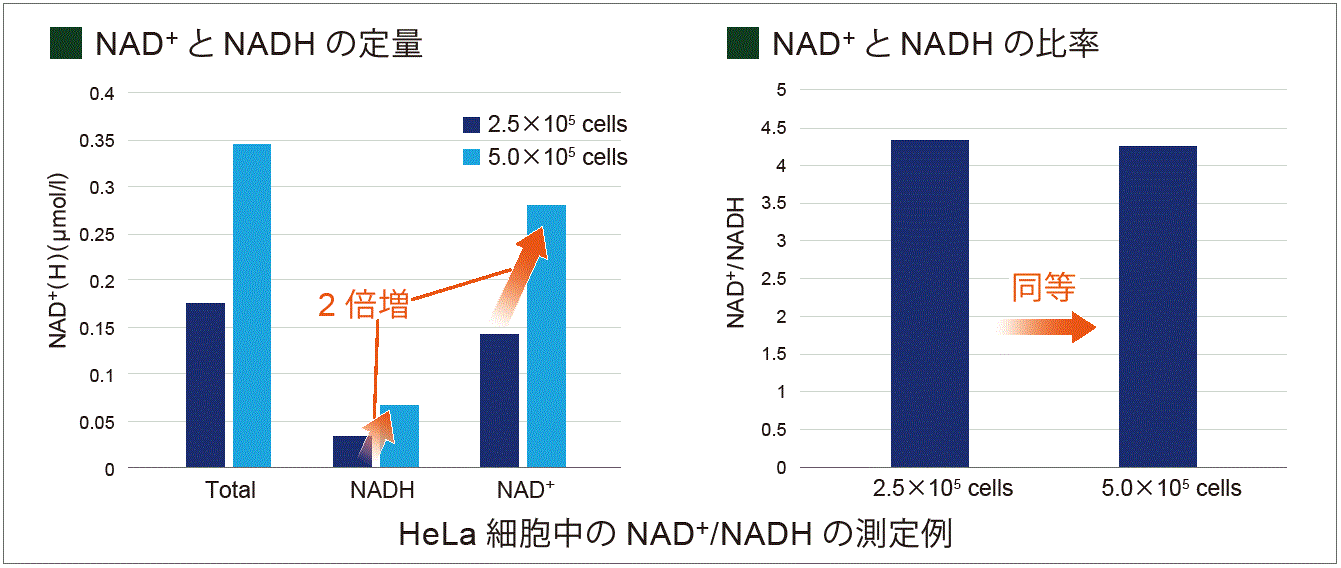
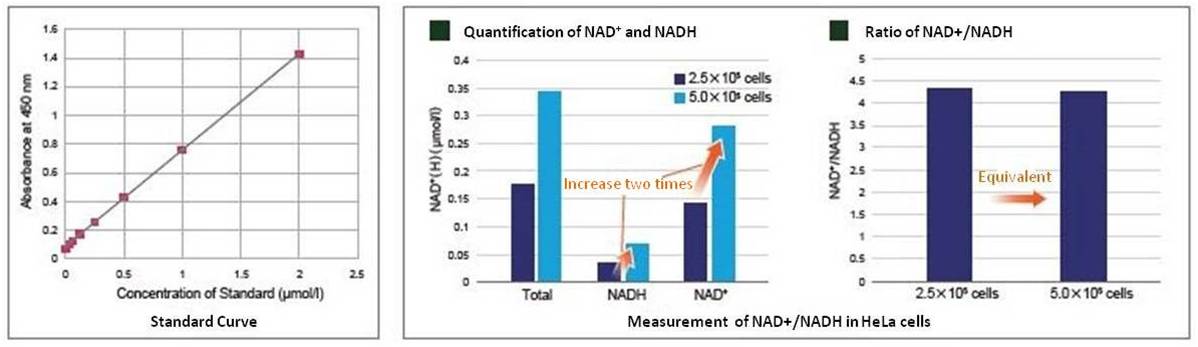
Standard curves were constructed using different concentrations of HeLa cells (2.5×105 and 5.0×105 cells) cultured in growth media. The standard curves were then used to determine the intracellular NAD+ and NADH levels. As a result, NAD+ and NADH levels varied depending on cell number while the change in cell number had no effect on the NAD+/NADH ratio.
Measurement of NAD+/NADH in Combination with Lactate Assay Kit
Change in metabolic activity was observed when the glycolytic inhibitor 2-Deoxy-D-glucose was added to HeLa cells.
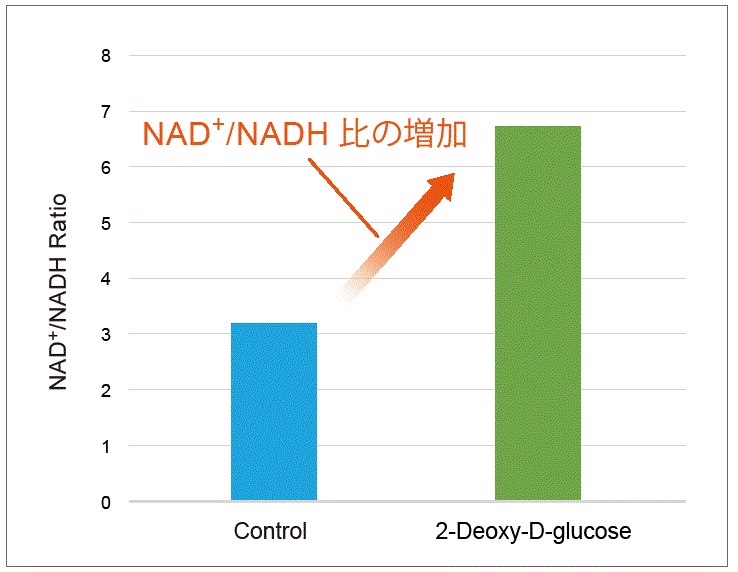
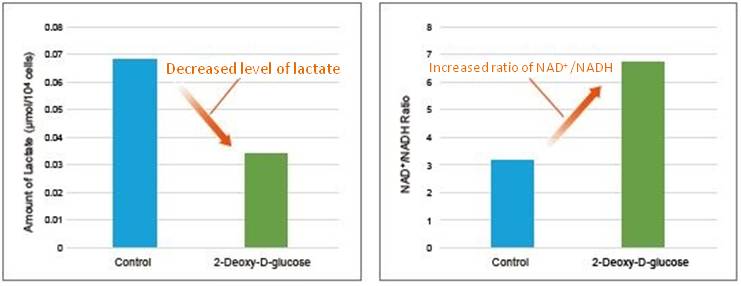
2-Deoxy-D-glucose was added to HeLa cells (1×106 cells) to obtain a final concentration of 6 mmol/l 2-Deoxy-D-glucose. After 24 hours of incubation, lactate levels in the supernatant were quantified using the Lactate Assay Kit-WST (Item#: L256), and the NAD+/NADH ratio was determined with the cell pellet after removing the supernatant using the NAD/NADH Assay Kit-WST.
As a result, intracellular glycolysis was inhibited by 2-Deoxy-D-glucose, which led to decreased lactate levels and an increase in the NAD+/NADH ratio.
Experimental Example: Change in Metabolism in Liver Tissue of NASH-Induced Mouse
It is known that non-alcoholic steatohepatitis (NASH) results in decreased ATP, α-ketoglutarate (α-KG), and NAD levels in affected tissues. ATP, α-KG, and NAD levels were measured in the liver tissue of type 1 diabetic model mice (STAM model) that were treated with a high-fat diet (NASH induction) since 4 weeks old. Measurement at 10 weeks old confirmed that ATP, α-KG, and NAD levels indeed decreased in the tissue samples after NASH induction.
Note: for more details about the experimental procedure, please refer to Q&A "Are there any examples of experiments using tissue samples?"
・Cellular ATP :ATP Assay Kit-Luminescence (Code:A550).
・Cellular α-KG:α-Ketoglutarate Assay Kit-Fluorometric (Code:K261).
・Cellular NAD :NAD/NADH Assay Kit-WST (Code:N509).
References:
| ATP | Francesco Bellanti, et al., "Synergistic interaction of fatty acids and oxysterols impairs mitochondrial function and limits liver adaptation during nafld progression", Redox Biology, 2018, 15, 86-96. |
|---|---|
| α-KG | Jianjian Zhao, et al., "The mechanism and role of intracellular α-ketoglutarate reduction in hepatic stellate cell activation", Bioscience Reports, 2020, 40, (3). |
| Ali Canbay, et al., "L‑Ornithine L‑Aspartate (LOLA) as a Novel Approach for Therapy of Non‑alcoholic Fatty Liver Disease", Drugs, 2019, 79, 39-44. | |
| NAD | Jinhan He, et al., "Activation of the Aryl Hydrocarbon Receptor Sensitizes Mice to Nonalcoholic Steatohepatitis by Deactivating Mitochondrial Sirtuin Deacetylase Sirt3", Mol. and Cell. Biol., 2013, 33, (10), 2047-55. |
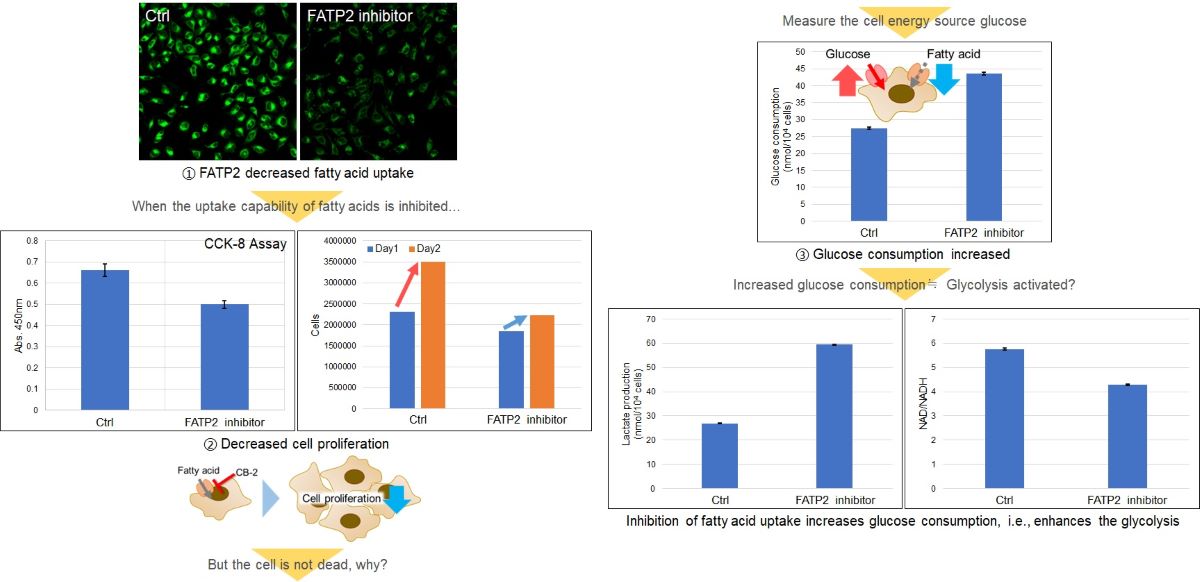
Application Data: Alteration of intracellular metabolism of HeLa cells by treatment with fatty acid transporter inhibitors
Fatty acids are important for membrane synthesis and are essential for cell proliferation. HeLa cells were treated with a fatty acid transporter inhibitor, and then the cell proliferation and intracellular metabolism (glucose consumption, Lactate release, and NAD/NADH ratio) were tested.
The results showed a decrease in cell proliferation, an increase in glucose consumption and Lactate release, and a decrease in the intracellular NAD+/NADH ratio, indicating a shift in the metabolic pathway to glycolysis.
<Related Products>
Free Acid Uptake: Fatty Acid Uptake Assay Kit (Code: UP07)
Cell Proliferation: Cell Counting Kit-8 (Code: CK04)
Glucose: Glucose Assay Kit-WST (Code: G264)
Lactate: Lactate Assay Kit-WST (Code: L256)
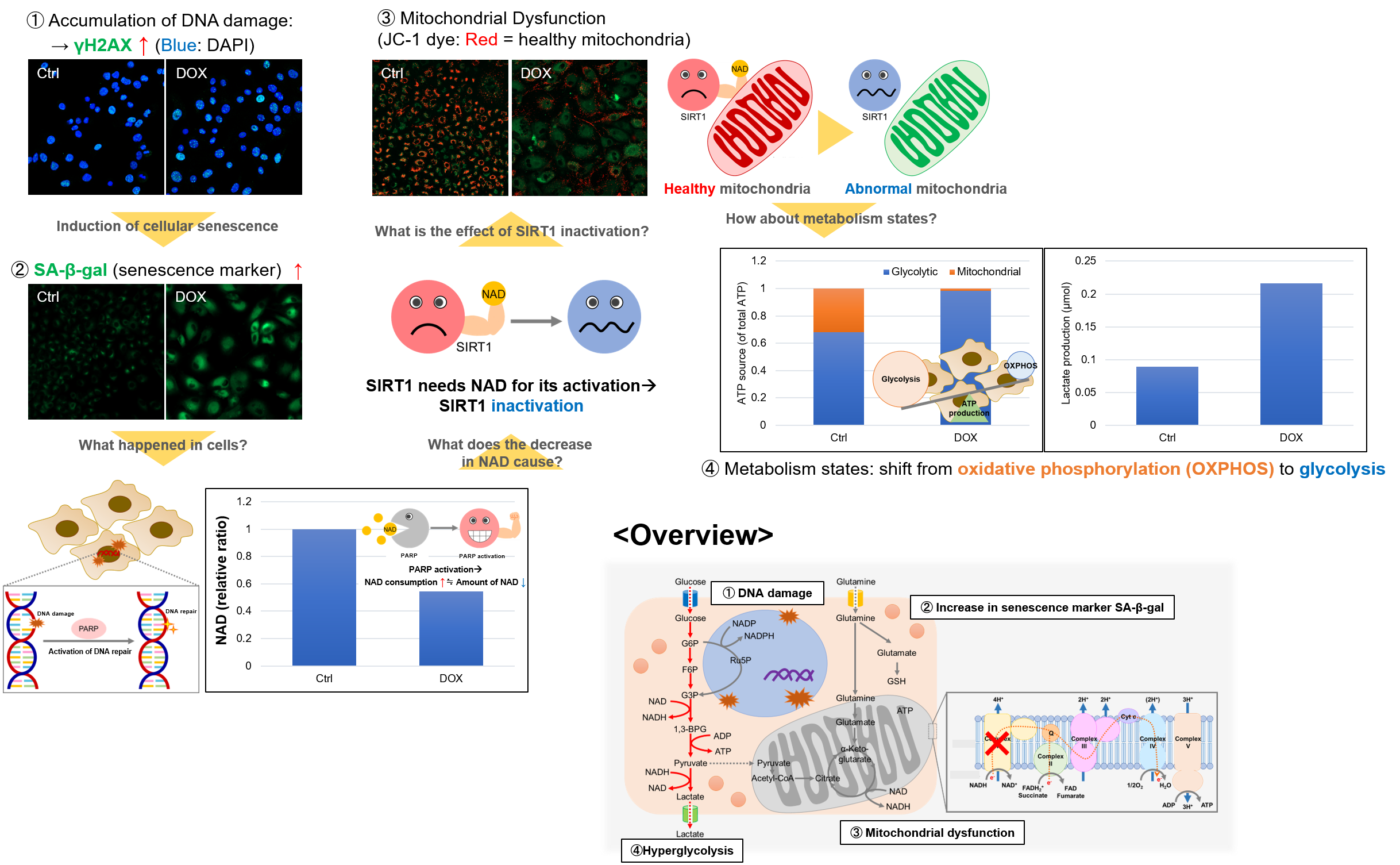
Experiment Example: Cellular Senescence with Metabolic shift
NAD(+) levels decline during the aging process, causing defects in nuclear and mitochondrial functions and resulting in many age-associated pathologies*. Here, we try to redemonstrate this phenomenon in the doxorubicin (DOX)-induced cellular senescence model with a comprehensive analysis of our products.
*S. Imai, et al., Trends Cell Biol, 2014, 24, 464-471
Related Products
| Category | Product | Unit | SKU |
|---|---|---|---|
|
Cellular Metabolism |
100 tests |
N509-10 |
|
| Lactate Assay Kit-WST | 50 / 200 tests | L256-10 / L256-20 | |
| Glycolysis/OXPHOS Assay Kit NEW | 50 tests | G270-10 | |
|
Cellular Senescence Detection |
1 / 3 / 10 plates |
SG04-01 / SG04-03 / SG04-10 |
|
|
Mitochondrial Membrane Potential |
10 set |
MT09-10 |
References
| ReferenceNo. | Sample | Citations |
|---|---|---|
| 1) | Organoids (Intestinal epithelial organoids in old mice) |
R. Uchida, Y. Saito, K. Nogami, Y. Kajiyama, Y. Suzuki, Y. Kawase, T. Nakaoka, T. Muramatsu, M. Kimura and H. Saito , "Epigenetic silencing of Lgr5 induces senescence of intestinal epithelial organoids during the process of aging", NPJ Aging Mech Dis., 2018,doi:10.1038/s41514-018-0031-5. |
| 2) | Adult T-cell leukemia cell line | T. Kozako, A. Aikawa, T. Ohsugi, Y. Uchida, N. Kato, K. Sato, K. Ishitsuka, M. Yoshimitsu and S. Honda, "High expression of NAMPT in adult T-cell leukemia/lymphoma and anti-tumor activity of a NAMPT inhibitor', Eur. J. Pharmacol.., 2019, 865, 172738. |
| 3) | Sirt 7 knockout fibroblasts | S. U. Sobuz, Y. Sato, T. Yoshizawa, F. Karim, K. Ono, T. Sawa, Y. Miyamoto, M. Oka and K. Yamagata, "SIRT7 regulates the nuclear export of NF-κB p65 by deacetylating Ran.', Biochim Biophys Acta Mol Cell Res.., 2019, 1866(9), 1355. |
| 4) | Human iPS-derived neural cells | K. Hayakawa, K. Nishitani and S. Tanaka, "Kynurenine, 3-OH-kynurenine, and anthranilate are nutrient metabolites that alter H3K4 trimethylation and H2AS40 O-GlcNAcylation at hypothalamus-related loci.", Sci Rep, 2019, 9(1), 19768. |
| 5) | HuH-7 | M. Mikeli, M. Fujikawa, K. Nagahisa, S. Yasuda, N. Yamada and T. Tanabe, "Contribution of GPD2/mGPDH to an alternative respiratory chain of the mitochondrial energy metabolism and the stemness in CD133-positive HuH-7 cells.", Genes Cells, 2020, 25(2), 139. |
| 6) | HeLa | J.Thapaa, K. Hashimoto, S. Sugawara, R. Tsujikawa, T. Okubo, S. Nakamura and H.Yamaguchi, "Hypoxia promotes Chlamydia trachomatis L2/434/Bu growth in immortal human epithelial cells via activation of the PI3K-AKT pathway and maintenance of a balanced NAD+/NADH ratio", Microbes Infect., 2020, DOI:10.1016/j.micinf.2020.04.010. |
Q & A
-
Q
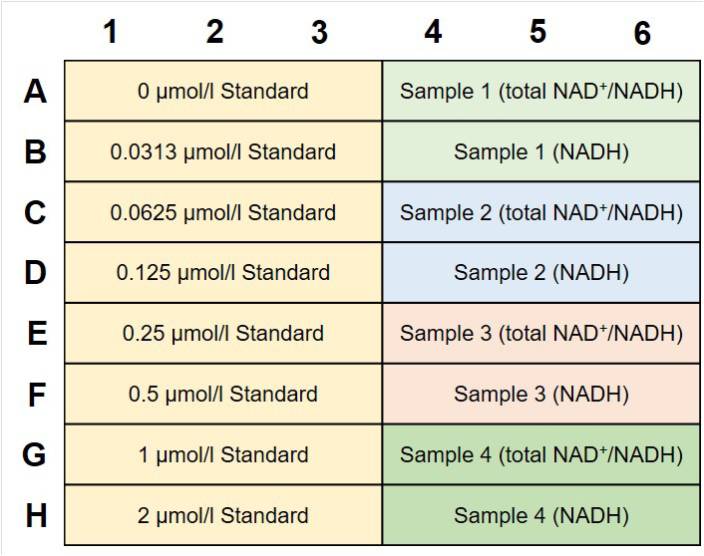
How many samples can I measure?
-
A
The number of samples that can be measured when the standard sample is serially diluted from 2 μmol / l is shown in the table above.
* The number of samples that can be recorded when the sample is measured in triplicates.It is necessary to create a calibration curve per measurement when you perform the measurement separately. If the measurement is done separately, the above sample number will be less.
-
Q
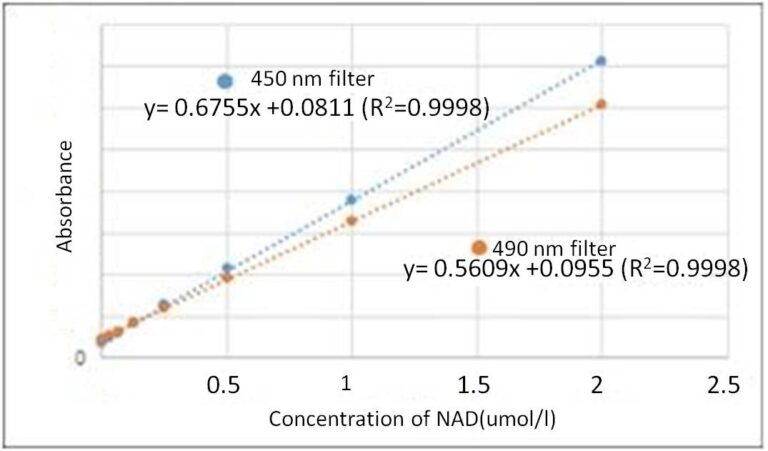
I do not have a 450 nm filter. What other filters can I use?
-
A
You can use filters with an absorbance between 450 nm and 490 nm. However, the absorbance value can become lower if the sample is not measured at 450 nm.
-
Q
Can I purchase the filtration tube separately?
-
A
No, we don’t sell the filtration tube included in the kit separately. If you need additional supplies, you can also use a commercially available filtration tube.
Supplier:Pall Corporation
Product:Nanosep® MF Centrifugal Devices (MWCO:10K, color:blue)
-
Q
How stable is Working solution?
-
A
You can’t store the Working solution. Please prepare the Working solution prior to use. Please protect from light because the Working solution ins light-sensitive. The Working solution is stable for 4 hours at room temperature with protection from light.
-
Q
Our samples did not change in color, are there any reasons for this?
-
A
NAD level contained in the sample may be lower than the detection limit that can be measured using this kit.
Please increase the number of cells or lower the dilution ratio if you dilute the sample.
-
Q
Are there any examples of experiments using tissue samples?
-
A
A.Yes. We have examples using mouse liver tissue.
General protocol is as below.
Extraction of NAD from liver samples via alkaline extraction method
1)Add cold 0.5 mol/l KOH solution to mouse liver (500 µl /100 mg).
*Make sure blood is properly removed from the tissue sample, using hemoperfusion or other methods.
*Any residual blood may affect the measurement.
2) Homogenize the tissue with Dounce tissue grinder.
3) Collect the sample and transfer it to a sample tube.
Wash the tissue grinder with 500 µl of cold 0.5 mol/l KOH solution and transfer the solution into the sample tube. (Total volume: 1 ml)
4) Add 1 ml of cold ultrapure water to the sample tube, mix well, and put it on ice for 5 minutes. (Total volume: 2 ml)
*If the solution is highly viscous, it may be difficult to separate after centrifugation in the next step.
In this case, attach a needle (about 25 g) to a syringe and use the syringe to pipette the sample solution about 20-30 times until the
sample solution can be smoothly pipetted within the syringe.
5) Centrifuge at 12,000 x g , 4 °C for 5 minutes and collect each 900 µl of the supernatant in two new tubes.
Preparation of sample for NAD/NADH measurement
*Continuing after “General Protocol, 1. Sample preparation, step (5)” in product manual.
*Prepare a mixture of 0.5 mol/l KOH solution and 1 mol/l KH2PO4 solution at a ratio of 9:5 as a dilution solution in advance.
6) Transfer 450 µl of the sample prepared in step (5) into an MWCO 10K filtration tube and centrifuge it at 15,000 x g for 20 minutes.
*If there is no more than 200 µl of solution left after centrifugation, extend the centrifugation time.
7) Transfer each 100 µl of the obtained filtrate into two 1.5 ml microtubes to make measurement samples for the NADH amount and the total NAD+/NADH amount.
8) Incubate the NADH amount measurement sample at 60 °C for 60 min, and cool the sample to room temperature.
9) Add 22 µl of 1 mol/l KH2PO4 solution to each tube containing the prepared total NAD+/NADH or NADH measurement sample.
Then, add 78 µl of dilution solution (mixture of KOH and KH2PO4) to each tube and mix well to make a measurement sample (200 µl in total).
*The extracted tissue samples cannot be stored. Please measure the sample within a day of the extraction.
*For dilution of the “standard” and “sample”, use a mixture of 0.5 mol/l KOH solution and 1 mol/l KH2PO4 solution at a ratio of 9:5 as the dilution solution.
NAD Level Changes in the Liver Tissue of NASH-Induced Mice
Handling and storage condition
| 0-5°C, Protect from light, 3.Protect from moisture | |
|
Danger / harmful symbol mark |
  
|
|---|---|















