Since a new coronavirus (SARS-CoV-2) was reported in Wuhan, China, in the winter of 2019, it has been mutating over and over, creating new variants that still threaten people’s lives. The scary thing about SARS-CoV-2 is the massive release of cytokines (cytokine storm) resulting from the viral infection, rather than the viral infection itself.
This cytokine storm causes the derailing of immune cell networks and results in severity, such as thrombosis and lung disease.1) Here, we would like to introduce you to recent studies on the relationship between senescent cells and SARS-CoV-2.
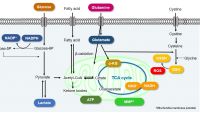
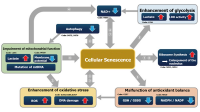
Senescent cells are cells in a state of irreversible growth arrest. Although it has long been thought to be preventing abnormal or DNA-damaged cells from proliferating and thus serves as a carcinogenesis prevention mechanism, it has also been discovered that senescent cells acquire SASPs (senescence-associated secretory phenotypes) and secrete factors that promote carcinogenesis in surrounding cells. Cellular senescence is indeed a double-edged sword.
According to the study by Lee et al., like other viruses, SARS-CoV-2 also induces cellular senescence as a stress response of infected cells. The group also hypothesized that SASP may amplify the inflammatory response to SARS-CoV-2 infection, then increase the risk of cytokine storm, pulmonary thrombosis, and so on.1) Camell et al. found that treatment with the senolytic fisetin in old mice infected with SARS-CoV-2 reduced mortality by 50%, reduced expression of inflammatory proteins in serum and tissues, and improved the immune response.2) These studies provide hope to people under the pandemic: senolytics (drugs that can eliminate senescent cells) can not only improve a healthy life span by eliminating senescent cells but can also be effective against SARS-CoV-2 infection.
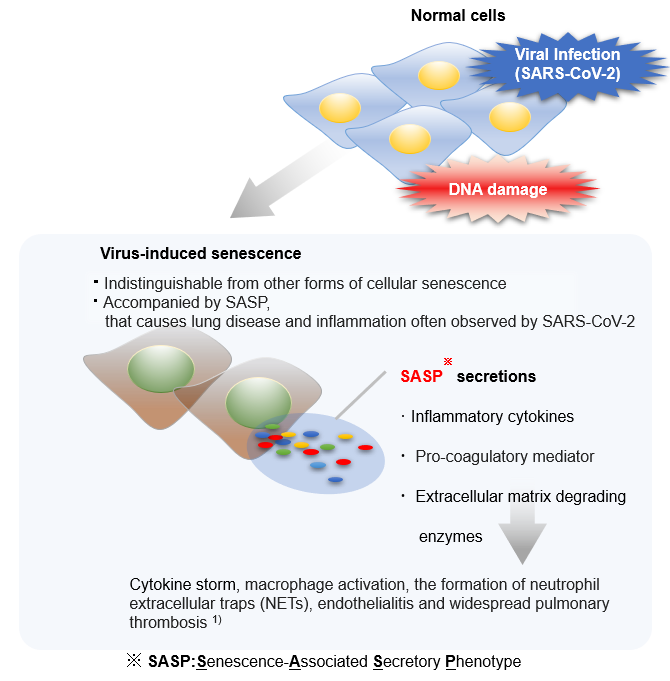
1. Lee, S., Yu, Y., Trimpert, J. et al. Virus-induced senescence is a driver and therapeutic target in COVID-19. Nature 599, 283–289 (2021).
Due to recent new findings, cellular senescence research has received further attention. Ever since it has been reported, the removal of senescent cells senescence can prolong a healthy lifespan. Among them the research and development of a senolytic drug, a drug that can remove senescent cells, which has now been getting very popular.
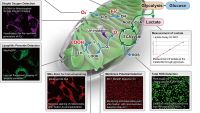
One of the interesting studies is that of Amor et al. They reported a new senolytic method using CAR T cells. In this study, Amor et al. identified the cell-surface protein that is broadly upregulated in senescent cells, but not in vital tissues. They also identified antigen “urokinase-type plasminogen activator receptor (uPAR).” They constructed uPAR-specific CAR T cells and treated them with a CCl4-induced liver fibrosis mouse model and a NASH induced liver fibrosis mouse model. As a result, without significant toxicity, the treated mouse models have less senescence marker “SA-ß-gal”, and reduction in liver fibrosis, which contributes to liver function compared with non-treated mouse models. The results indicate that not only the removal of senescent cells by CAR T cells is possible, but also senolytic CAR T cells can work effectively to reduce liver fibrosis.3)
Another interesting research is done by Suda et al. They found GPNMB (glycoprotein nonmetastatic melanoma protein B), which they identified as a molecular target for senolytic therapy. It is a molecule with a transmembrane domain that was enriched in senescent cells (seno-antigen). In their study, they vaccinated the mouse with GPNMB and observed individual senescence and systemic metabolic abnormalities. They showed that the reduction of GPNMB-positive genes suppresses senescence.4) This suggests that vaccination targeting the seno-antigen may also be a strategy for senolytic therapy.
Although the onslaught of SARS-CoV-2 is likely to continue for some time, the development of vaccines and therapeutic drugs is progressing. In addition, new discoveries have been made through the latest research on senolytics. They have attracted attention not only as a health-promoting drug, but also as a therapeutic drug, and we can expect further development of this research in the future.
1. Lee, S., Yu, Y., Trimpert, J. et al. Virus-induced senescence is a driver and therapeutic target in COVID-19. Nature 599, 283–289 (2021).
2. Camell, C. D. et al. Senolytics reduce coronavirus-related mortality in old mice. Science 373, eabe4832 (2021).
3. Amor, C. et al., Senolytic CAR T cells reverse senescence-associated pathologies, Nature 583, 127–132 (2020).
4. Suda, M., Shimizu, I., Katsuumi, G. et al. Senolytic vaccination improves normal and pathological age-related phenotypes and increases lifespan in progeroid mice. Nat Aging 1, 1117–1126 (2021).