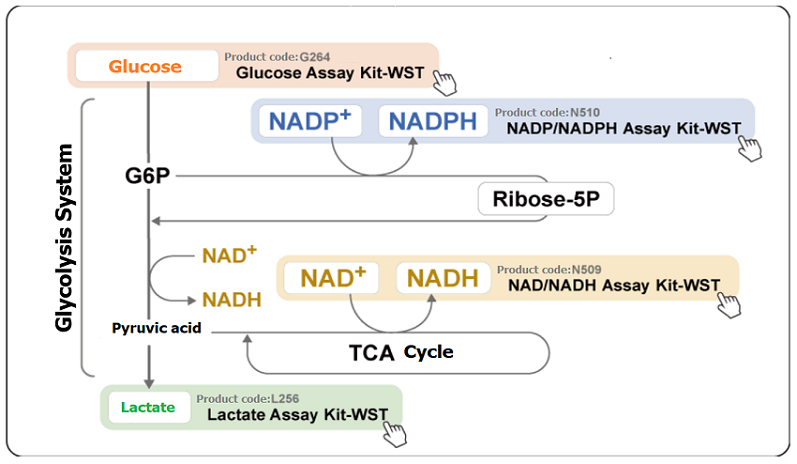
Analyzing the various intracellular metabolisms i.e., the glycolysis system, the tricarboxylic acid (TCA) cycle, and the electron transport chain, is important when trying to understand cellular states. Metabolites and energy sources [e.g. glucose, lactate, and NAD (P)/NAD (P) H] are the indicators used for the analysis of intracellular metabolisms.
<Case examples Selected to Illustrate the Associations between Metabolism and Disease>
Intracellular metabolic states are often identified in the assessment of disorder models (e.g. cancer and diabetes) and mitochondrial function. The table shown below represents the case examples that used lactate levels and NAD+/NADH ratio as indicators for their assessments.
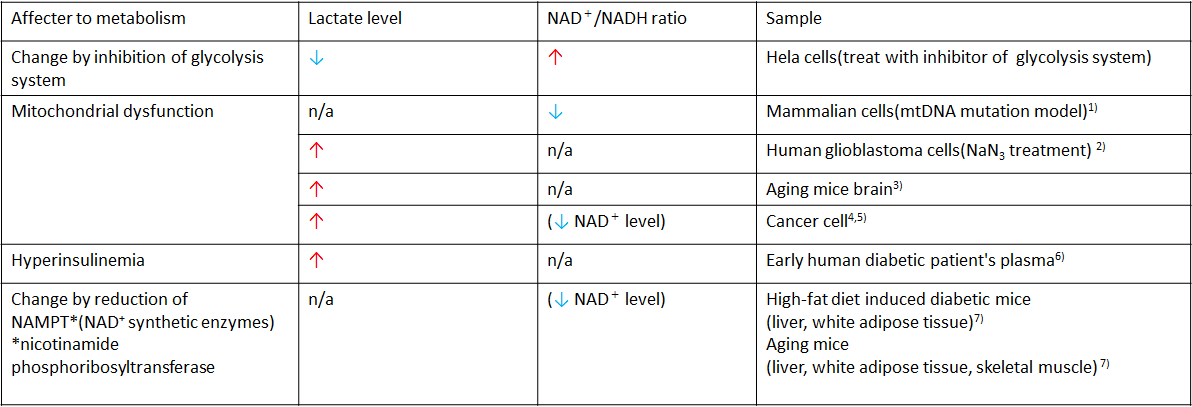
In recent years, the association between NAD+ and age-related disorders has received a lot of attention. Mouse models of aging showed that decreased levels of NAD+ in the liver indicates that the inhibition of NAD+ synthetic enzymes triggers aging-like deterioration of cellular function. Additionally, there is a strong correlation between the degradation of mitochondrial function and the decline of NAD+ levels1), 11). These changes will eventually lead to aging-like deterioration of cellular function.1) The reduction in mitochondrial function leads to high levels of lactate which is closely related to the progression of aging-related disorders, such as cancer and diabetes. Therefore, analyzing changes in NAD+ and lactate levels is important to understanding their relation to aging.
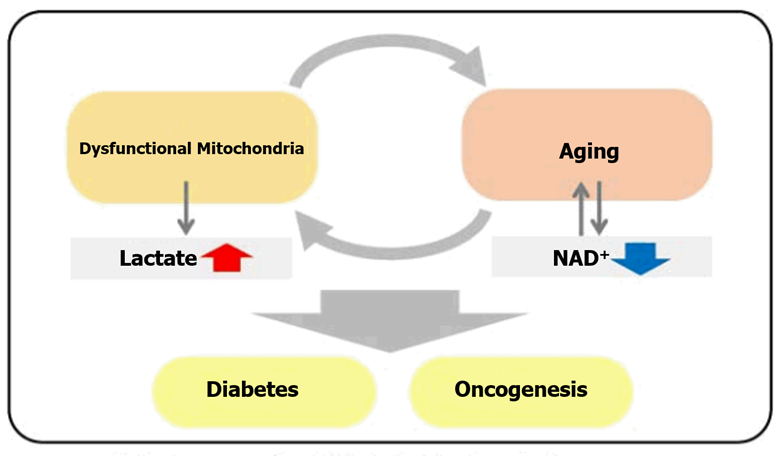
<Metabolic Pathways: DNA Damage-Induced Senescent Cells>
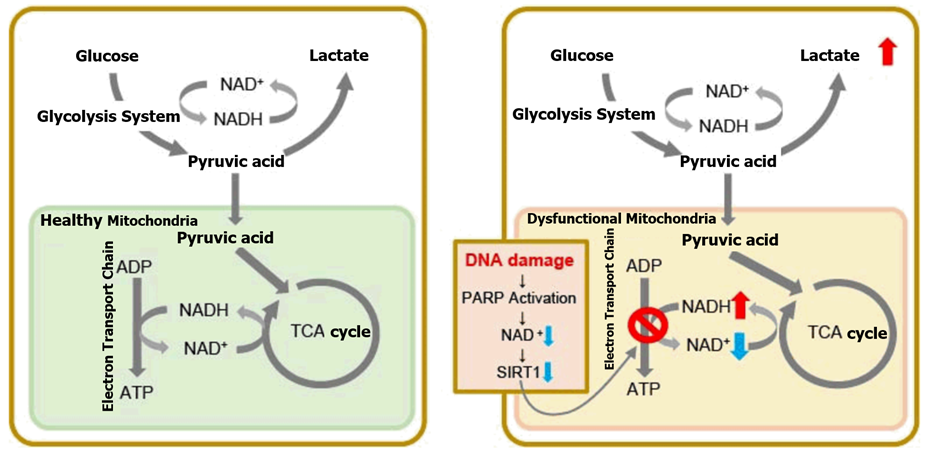
- With deteriorating mitochondrial function, senescent cells mainly produce ATP through anaerobic glycolysis, which increases lactate production.
- DNA damage is one of the causes of aging-induced deterioration of mitochondrial function. When DNA accumulates damage, DNA repair mechanisms are activated, increasing NAD+ consumption.
- Sirtuin 1 (SIRT1) activity is an important factor in maintaining mitochondrial function. Decreased levels of NAD+ reduce Sirtuin 1 (SIRT1) activity which leads to the deterioration of mitochondrial function (Inhibition of the electron transport chain→ATP production and decreased levels of NAD+).
Reference
1) A. Bratic and N. Larsson, “The role of mitochondria in aging”, J Clin Invest. 2013, 123(3), 951.
2) M. E. Beckner, G. T. Gobbel, R. Abounader, F. Burovic, N. R Agostino, J. Laterra and I. F. Pollack, “Glycolytic glioma cells with active glycogen synthase are sensitive to PTEN and inhibitors of PI3K and gluconeogenesis”, Laboratory Investigation, 2005, 85(12), 1457.
3) J. M. Ross, J. Oberg, S. Brene, G. Coppotelli, M. Terzioglu, K. Pernold, M. Goiny, R. Sitnikov, J. Kehr, A. Trifunovic, N. Larsson, B. J. Hoffer, and L. Olson, “High brain lactate is a hallmark of aging and caused by a shift in the lactate dehydrogenase A/B ratio”, PNAS, 2010, 107(46), 20087.
4) A. Garrido and N. Djouder, “NAD+ Deficits in Age-Related Diseases and Cancer”, Trends Cancer, 2017, 3(8), 593.
5) I. San-Millancorresponding and G. A. Brooks, “Reexamining cancer metabolism: lactate production for carcinogenesis could be the purpose and explanation of the Warburg Effect”, Carcinogenesis, 2017, 38(2), 119.
6) Y. Wu, Y. Dong, M. Atefi, Y. Liu, Y. Elshimali, and J. V. Vadgama, “Lactate, a Neglected Factor for Diabetes and Cancer Interaction”, Mediators Inflamm., 2016. DOI: 10.1155/2016/6456018.
7) J. Yoshino, K. F. Mills, M. J. Yoon and S. Imai, “Nicotinamide mononucleotide, a key NAD+ intermediate, treats the pathophysiology of diet- and age-induced diabetes in mice”, Cell Metab., 2011, 14(4), 528.
8)L. R. Stein and S. Imai, “Specific ablation of Nampt in adult neural stem cells recapitulates their functional defects during aging”, EMBO J., 2014, 33(12), 1321.
9) Z. Feng, R. W. Hanson, N. A. Berger and A. Trubitsyn, “Reprogramming of energy metabolism as a driver of aging”, Oncotarget., 2016, 7(13), 15410.
10) S. Imai and L. Guarente, “NAD+ and sirtuins in aging and disease”, Trends in Cell Biology, 2014, 24(8), 464.
11) J. Wu, Z. Jin, H. Zheng and L. Yan, “Sources and implications of NADH/NAD+ redox imbalance in diabetes and its complications”, Diabetes Metab. Syndr. Obes., 2016, 9, 145.




