Maleimido-C3-NTA

Cross-Linker
-
Product codeM035 Maleimido-C3-NTA
-
CAS No.869843-95-8
-
Chemical nameN-[5-(3'-Maleimidopropylamido)-1-carboxypentyl]iminodiacetic acid, disodium salt, monohydrate
-
MWC18H23N3Na2O9・H2O=489.38
| Unit size | Price | Item Code |
|---|---|---|
| 10 mg | M035-10 |
Product Description
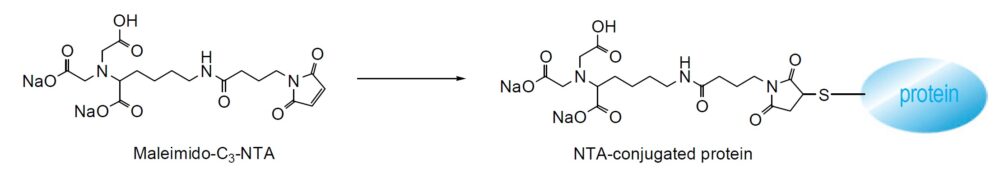
Maleimido-C3-NTA is utilized to modify the surfaces on which thiol groups are attached. Through the NTA moiety attached to the surface, genetically expressed protein, bearing a hexahistidine extension at its terminus, can be immobilized via Ni (II) (His-Tag method). Using this technique, Dr. Noji and co-workers were able to directly observe the rotation of F1-ATPase with a fluorescence microscope.
Chemical Structure
References
1) E. Hochuli, H. Doeli and A. Schacher, "New Metal Chelate Adsorbent Selective for Proteins and Peptides Containing Neighbouring Histidine Residues", J. Chromatogr., 1987, 411, 177.
2) E. Hochuli, "Large-Scale Chromatography of Recombinant Proteins", J. Chromatogr., 1988, 444, 293.
3) Y. C. Sasaki, Y. Suzuki and T. Ishibashi, "Fluorescent X-ray Interference from a Protein Monolayer", Science, 1994, 263, 62.
4) G. B. Sigal, C. Bamdad, A. Barberis, J. Strominger and G. M. Whitesides, "A Self-Assembled Monolayer for The Binding and Study of Histidine-Tagged Proteins by Surface Plasmon Resonance, Anal. Chem., 1996, 68, 490.
5) E. L. Schmid, T. A. Keller, Z. Dienes and H. Vogel, "Reversible Oriented Surface Immobilization of Functional Proteins on Oxide Surface", Anal. Chem., 1997, 69, 1979.
Handling and storage condition
| Appearance: | White pale yellowish-white powder |
|---|---|
| Purity (HPLC): | ≥97.0% (HPLC) |
| Solubility in water: | To pass test (clear, colorless) |
| IR spectrum: | Authentic |
| NMR spectrum: | Authentic |
| 0-5°C, Protect from moisture |








