Cell Counting Kit-F

Cell Proliferation / Cytotoxicity Assay
-
Product codeCK06 Cell Counting Kit-F
| Unit size | Price | Item Code |
|---|---|---|
| 500 tests | CK06-10 |
| 500 tests | -Calcein-AM DMSO solution- | 110 μl x1 |
|---|
Product Description
Cell Counting Kit-F(CCK-F) is utilized for the fluorometric determination of living cell numbers. The amount of the fluorescent dye, calcein, hydrolyzed by esterases in cells is directly proportional to the number of viable cells in culture media (Fig. 1). Since esterases and phenol red in the culture medium interfere with the fluorescence measurement, replacing the cell culture medium with PBS is necessary prior to adding the Calcein-AM assay solution. The excitation and the emission wavelengths of calcein are 485 nm and 535 nm, respectively (Fig. 3). An incubation of 10 to 30 minutes gives sufficient fluorecence intensity for the cell viability determination.
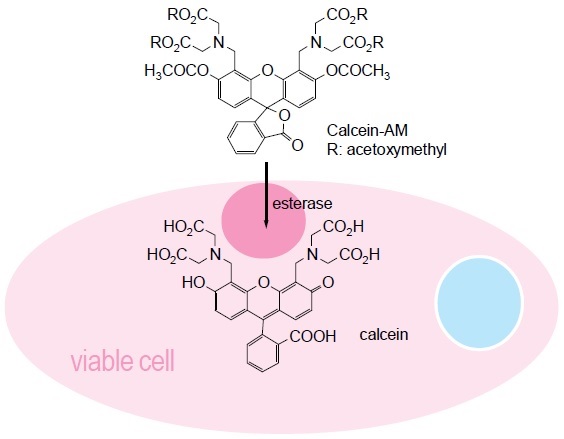
Fig. 1 Cell viability detection mechanism with CCK-F
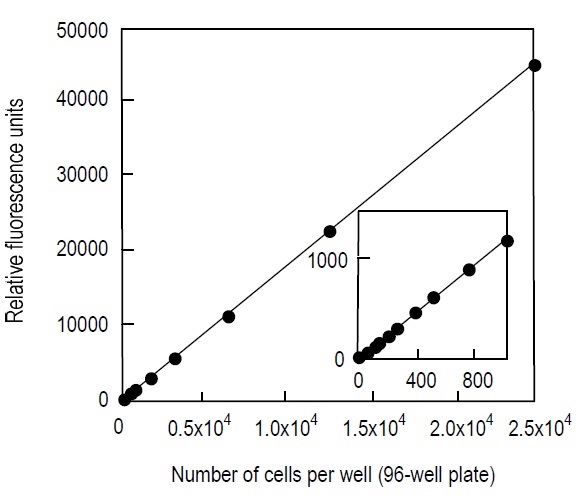
Fig. 2 Typical calibration curve using CCK-F
Cell line: HL60
Culture medium: RPMI1640, 10% FCS, L-glutamine
Incubation: 37 °C, 5% CO2, 30 min.
Detection: 485 nm excitation, 535 nm emission
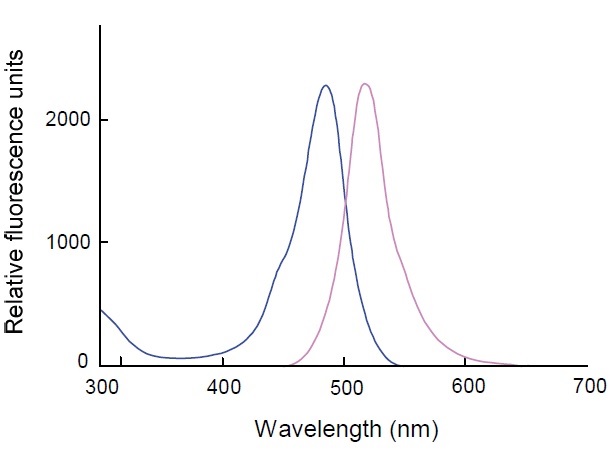
Fig. 3 Fluorescence spectrum of Calcein/ PBS solution, pH 7.4
Manual
References
1. N. G. Papadopoulos, et al., An improved fluorescence assay for the determination of lymphocyte-mediated cytotoxicity using flow cytometry. J Immunol Methods. 1994;177:101-111.
2. S. Sakamoto, et al., Increased Expression of CYR61, an Extracellular Matrix Signaling Protein, in Human Benign Prostatic Hyperplasia and Its Regulation by Lysophosphatidic Acid. Endocrinology. 2004;145:2929-2940.
3. T. Uruma, et al., Chlamydia pneumoniae growth inhibition in human monocytic THP-1 cells and human epithelial HEp-2 cells by a novel phenoxazine derivative. J Med Microbiol. 2005;54:1143-1149.
4. B. Ashibe, et al., Dual Subcellular Localization in the Endoplasmic Reticulum and Peroxisomes and a Vital Role in Protecting against Oxidative Stress of Fatty Aldehyde Dehydrogenase Are Achieved by Alternative Splicing. J Biol Chem. 2007;282:20763-20773.
5. Z. Bai, et al., CXC Chemokine Ligand 12 Promotes CCR7-Dependent Naive T Cell Trafficking to Lymph Nodes and Peyer’s Patches. J Immunol. 2009;182:1287-1295.
Q & A
-
Q
How many cells should there be in a well?
-
A
At least 50 cells are necessary per well (100 μl medium) when using a standard 96-well plate. However, we recommend using at least 1,000 cells per well for more reliable and consistent data.
-
Q
Does calcein stain viable cells?
-
A
Yes, calcein stays inside of the cell. The viable cell can be visualized using a fluorescent microscope. If a longer observation period is necessary, please try CFSE (Product Code: C375-10).
-
Q
Does phenol red affect the assay?
-
A
Yes. A washing process is required prior to adding assaysolution to the plate. Wells with only PBS also have a rather high fluorescent background, so it will be necessary to subtract the background fluorescence from the total fluorescence.
-
Q
Can I use this kit for bacterial cell counting?
-
A
No. Calcein-AM cannot pass through bacterial cell walls, so they cannot be stained using this kit.
-
Q
Is the CCK-F assay solution stable?
-
A
No. Calcein-AM in CCK-F is easily hydrolyzed in PBS. Prepare only the required volume of assay solution for the experiment.
-
Q
Is there a correlation between CCK-F and the thymidine incorporation assay?
-
A
Yes. However, please note that since CCK-F uses a different assay mechanism from that of the thymidine assay, results may differ. Comparison data are shown in the technical manual, which is available at www.dojindo.com/tm.
-
Q
Is CCK-F assay solution toxic to cells?
-
A
Since the assay solution is prepared with PBS, some cells may be affected by PBS. Also, calcein binds calcium ions in the cell, so the reduction of the free calcium ions causes some damage to cell functions.
-
Q
What other filters can I use?
-
A
You can use excitation filters between 460 nm and 490 nm and emission filters between 510 nm and 540 nm.
-
Q
Can I use CCK-F for 384-well plates?
-
A
CCK-F can be used for 384-well plates by adding 5 μl (instead of 10 μl) CCK-F assay solution to 50 μl PBS solution per well.
Handling and storage condition
| -20°C, Protect from light | |
|
Danger / harmful symbol mark |

|
|---|---|














