BCECF-AM

Reagent for Monitoring Intracellular pH
-
Product codeB262 BCECF-AM
-
CAS No.117464-70-7
-
Chemical name3'-O-Acetyl-2',7'-bis(carboxyethyl)-4 or 5-carboxyfluorescein, diacetoxymethyl ester
-
MWC35H28O15=688.59
| Unit size | Price | Item Code |
|---|---|---|
| 1 mg | B262-10 |
Product Description
BCECF is the most widely used intracellular pH probe. Dr. Tsien and others improved this carboxyfluorescein by introducing two extra carboxylates that allow it to be retained better by the cell. BCECF is highly water-soluble because it has 4 to 5 negative charges at neutral pH; it becomes difficult to pass through the cell membrane after loading. Its pKa value, 6.97, is higher than that of carboxyfluorescein. BCECF has an isosbestic point at 439 nm in the excitation spectra, so it can be used in ratiometry, similar to Fura 2. Wavelengths of 505 nm and 439 nm are usually used for the ratiometric assay, and 490 nm and 450 nm filters are set in front of the excitation light source. The 530 nm filter is used for its fluorescent signal. Please note that the excitation spectrum is slightly different from the absorption spectra. BCECF-AM is an acetoxymethyl ester of BCECF that enables easy loading of BCECF into cells. BCECF-AM accumulates in a cell only by incubation as do the other acetoxymethyl esters. BCECF-AM is very sensitive to moisture; it should be carefully handled. The color of the DMSO solution changes from pale yellow to dark orange with decomposition of the AM form. Therefore, hydrolysis of the AM ester can be monitored by changes in color.
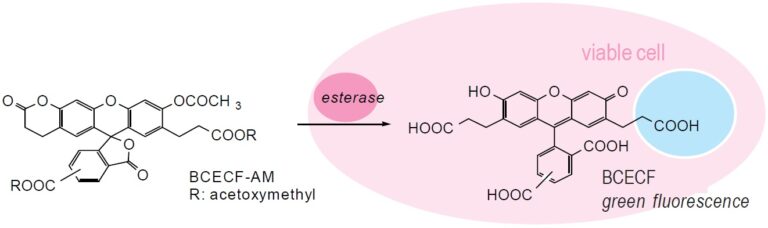
Fig. 1 Cell staining mechanism
Technical info
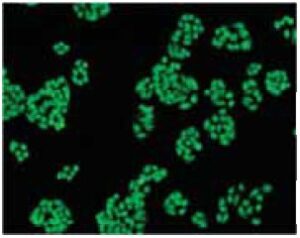
Fig. 2 Cell staining with BCECF-AM
Cell type: HeLa
General Protocol (for Human Neutrophil)*
Reagents:
– 1 mM BCECF-AM/DMSO solution (1 mg BCECF in 1.45 ml DMSO)
– HEPES buffer saline (20 mM HEPES, 153 mM NaCl, 5 mM KCl, 5 mM glucose, pH 7.4)
Protocol:
1. Suspend cells in HEPES buffer solution to prepare 4×107 cells per ml.
2. Add 1 mM BCECF-AM/DMSO solution to the cell suspension to prepare 3 μM BCECF-AM (1/300 vol of cell suspension) as the final concentration.
3. Incubate the cell suspension at 37ºC for 30 minutes.
4. Wash the cells 3 times with HEPES buffer saline and then prepare 3×106 cells per ml of the cell suspension.
5. Determine the fluorescence intensity using a fluorescence microscope or a confocal laser microscope coupled with an image analyzer.
* Cell staining conditions depend on cell type, so it is necessary to optimize the conditions for each experiment
References
1) R. A. Steinhardt and D. Mazia, "Development of K+-conductance and Membrane Potentials in Unfertilized Sea Urchin Eggs After Exposure to NH4OH", Nature, 1973, 241, 400.
2) T. J. Rink, R. Y. Tsien and T. Pozzan, "Cytoplasmic pH and Free Mg2+ in Lymphocytes", J. Cell Biol., 1982, 95, 189.
3) A. M. Paradiso, R. Y. Tsien and T. E. Machen, "Na+ -H+ Exchange in Gastric Glands as Measured with a Cytoplasmic-trapped, Fluorescent pH Indicator", Proc. Natl. Acad. Sci. USA, 1984, 81, 7436.
4) S. Grinstein, B. Elder and W. Furuya, "Phorbol Ester-induces Changes of Cytoplasmic pH in Neutrophils: Role of Exocytosis in Na+ - H+ Exchange", Am. J. Physiol., 1985, 248, C379.
5) G. B. Zavoico, E. J. Cragoe and M. B. Feinstein, "Regulation of Intracellular pH in Human Platelets", J. Biol. Chem., 1986, 261(28), 13160.
6) G. R. Bright, W. Fisher, J. Rogowska and L. Taylor, "Fluorescence Ratio Imaging Microscopy: Temporal and Spatial Measurements of Cytoplasmic pH", J. Cell Biol., 1987, 104, 1019.
7) C. Aalkjaer and E. J. Gragoe Jr, "Intracellular pH Regulation in Resting and Contracting Segments of Rat Mesenteric Resistance Vessels", J. Physiol., 1988, 402, 391.
8) K. Tsujimoto, M. Semadeni, M. Huflejt and L. Packer, "Intracellular pH of Halobacteria Can Be Determined by the Fluorescent Dye 2',7'-bis(carboxyethyl)-5(6)-carboxyfluorescein", Biochem. Biophys. Res. Commun., 1988, 155, 123.
9) M. A. Kolber, R. R. Quinones, R. E. Gress and P. A. Henkart, "Measurament of Cytotoxicity by Target Cell Release and Retention of the Fluorescent Dye Bis-carboxyethyl-carboxyfluorescein(BCECF)", J. Immunol. Methods, 1988, 108, 255.
10) H. Harada, Y. Kanai, M. Anzai and Y. Suketa, "cAMP Activates Cl-/HCO3- Exchange for Regulation of Intracellular pH in Renal Epithelial Cells", Biochim. Biophys. Acta, 1991, 1092, 404.
11) C. C. Freudenrich, E. Murphy, L. A. Levy, R. E. London and M. Lieberman, "Intracellular pH Modulates Cytosolic Free Magnesium in Cultured Chicken Heart Cells", Am. J. Physiol., 1992, 262(4), C1024.
12) K. Khodakhah and D. Ogden, "Functional Heterogeneity of Calcium Release by Inositol Triphosphate in Single Purkinje Neurones, Cultured Cerebellar Astorocytes, and Peripheral Tissues", Proc. Natl. Acad. Sci. USA, 1993, 90, 4976.
13) G. Boyarsky, C. Hanssen and L. A. Clyne, "Superiority of in vitro Over in vivo Calibrations of BCECF in Vascular Smooth Muscle Cells", FASEB J., 1996, 10, 1205.
14) S. A. Weston and C. R. Parish, "New Fluorescent Dyes for Lymphocyte Migration Studies Analysis by Flow Cytometry and Fluorescent Microscopy", J. Immunol. Methods, 1990, 133, 87.
15) L. S. D. Clerck, C. H. Bridts, A. M. Mertens, M. M. Moens and W. J. Stevens, "Use of Fluorescent Dyes in the Determination of Adherence of Human Leucocytes to Endothelial Cells and the Effects of Fluorochromes on Cellular Function", J. Immunol. Methods, 1994, 172, 115.
Handling and storage condition
| Appearance: | Pale orange to pale orange-brown powder or solid |
|---|---|
| Purity (HPLC): | ≧ 90.0 % |
| Fluorescence spectrum: | To pass test |
| NMR spectrum: | To pass test |
| -20°C, Protect from light, Protect from moisture |












