-Cellstain- Double Staining Kit

Cell Double Staining
-
Product codeCS01 -Cellstain- Double Staining Kit
| Unit size | Price | Item Code |
|---|---|---|
| 1 set | CS01-10 |
| 1 set | ・Solution A:Calcein-AM Stock Solution (1 mmol/l) Calcein-AM in DMSO ・Solution B:PI Stock Solution |
50 μl×4 vials |
|---|
Description
Product Description
-Cellstain-Double Staining Kit is utilized for simultaneous fluorescence staining of viable and dead cells. This kit contains Calcein-AM and Propidium Iodide (PI) solutions, which stain viable and dead cells, respectively (Fig. 1). Calcein-AM, an acetoxymethyl ester of calcein, is highly lipophilic and cell membrane permeable. Although Calcein-AM itself is not a fluorescent molecule, the calcein generated from Calcein- AM by esterase in a viable cell emits a strong green fluorescence (excitation: 490 nm, emission: 515 nm). Therefore, Calcein-AM only stains viable cells. On the other hand, PI, a nuclei staining dye, cannot pass through a viable cell membrane. It reaches the nucleus by passing through disordered areas of dead cell membrane and intercalates with the DNA double helix of the cell to emit red fluorescence (excitation: 535 nm, emmision: 617 nm). Since both calcein and PI-DNA can be excited with 490 nm, simultaneous monitoring of viable and dead cells is possible with a fluorescence microscope. With 545 nm excitation, only dead cells can be observed (Fig. 2). Since optimal staining conditions differ from cell line to cell line, we recommend that a suitable concentration of PI and Calcein- AM be individually determined. Please note that PI is suspected to be highly carcinogenic; careful handling is required.
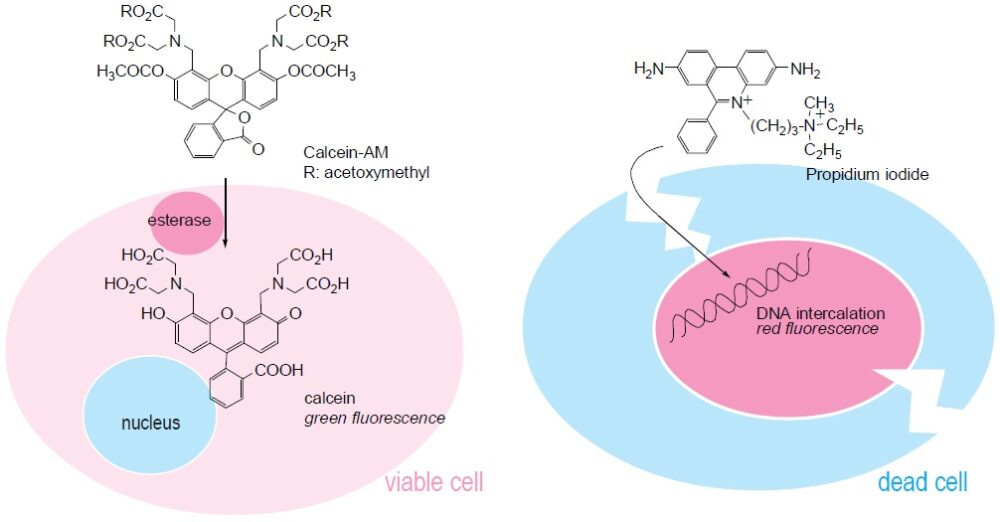
Fig. 1 Assay system to determine viable cells and dead cells
Manual
Technical info
Optimization Procedure
The following steps may be necessary to determine the suitable concentration of each reagent:
1. Prepare dead cells by 10 minutes incubation in 0.1% saponin or 0.1-0.5% digitonin or by 30 minutes incubation in 70% ethanol.
2. Stain dead cells with 0.1-10 μM PI solution to find a PI concentration that stains the nucleus only, not the cytosol.
3. Stain dead cells with 0.1-10 μM Calcein-AM solution to find a Calcein-AM concentration that does not stain the cytosol. Then stain viable cells with that Calcein-AM solution to check whether the viable cell can be stained.
Data
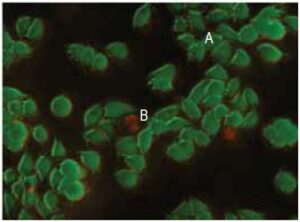
Fig. 2 Cell staining with Double Staining Kit HeLa cell, incubated with assay solution for 15 minutes.
A) viable cell
B) dead cell
References
1. L. S. D. Clerck, et al., Use of Fluorescent Dyes in the Determination of Adherence of Human Leucocytes to Endothelial Cells and the Effects of Fluorochromes on Cellular Function. J Immunol Methods. 1994;172:115-124.
2. E. S. Kaneshiro, et al., Reliability of Calcein Acetoxy Methyl Ester and Ethidium Homodimer or Propidium Iodide for Viability Assessment of Microbes. J Microbiol Methods. 1993;17:1-16.
3. N. G. Papadopoulos, et al., An Improved Fluorescence Assay for the Determination of Lymphocyte-Mediated Cytotoxicity Using Flow Cytometry. J Immunol Methods. 1994;177:101-111.
4. M. Adler, et al., Cytotoxic actions of the heavy metal chelator TPEN on NG108-15 neuroblastoma-glioma cells. Neurotoxicology. 1999;20:571-582.
5. P. G. Bush, et al., Viability and volume of in situ bovine articular chondrocytes-changes following a single impact and effects of medium osmolarity. Osteoarthritis Cartilage. 2005;13:54-65.
Handling and storage condition
| -20°C, Protect from light | |
|
Danger / harmful symbol mark |

|
|---|---|









