ACE Kit - WST

ACE Inhibitory Activity Assay
- Colorimetric Microplate Assay Possible
- Possible to Test Multiple Samples at One Time
- Simple Protocol
- No Harmful Organic Solvent Required
-
Product codeA502 ACE Kit - WST
| Unit size | Price | Item Code |
|---|---|---|
| 50 tests | A502-05 | |
| 100 tests | A502-10 |
| 50 tests | ・Substrate Buffer ・Enzyme A ・Enzyme B ・Enzyme C ・Coenzyme ・Indicator Solution |
1 ml x1 x1 x1 x1 x1 5 ml x1 |
|---|---|---|
| 100 tests | ・Substrate Buffer ・Enzyme A ・Enzyme B ・Enzyme C ・Coenzyme ・Indicator Solution |
1 ml x2 x2 x2 x2 x2 5 ml x2 |
Description
The kit is used for the determination of ACE inhibition activity. ACE works in the Renin-Angiotensin system, which is one of the mechanisms of blood pressure control, to convert Angiotensin I to the vasopressor Angiotensin II. This enzyme also contributes to elevated blood pressure due to its role in breaking down the antihypertensive peptide Bradykinin. In recent years, food and supplements containing ingredients that block ACE have received attention for their use in preventing high blood pressure. The conventional method of measuring ACE inhibition employs the synthetic substrate Hippuryl-His-Leu. Hippuric acid from the synthetic substrate is extracted with ethyl acetate, condensed, redissolved, and then read at an absorbance of 228 nm. This method is cumbersome and measurement is subjected to error due to residual ethyl acetate. ACE inhibition Assay Kit enzymatically detects 3-Hydroxybutyric acid (3HB), which is made from 3-Hydryoxybutyryl-Gly-Gly-Gly (3HB-GGG). Using a 96-well format, it is possible to test multiple samples at one time. In addition, there is no need to use harmful organic solvents, resulting in a safe, simple, and highly reproducible assay.
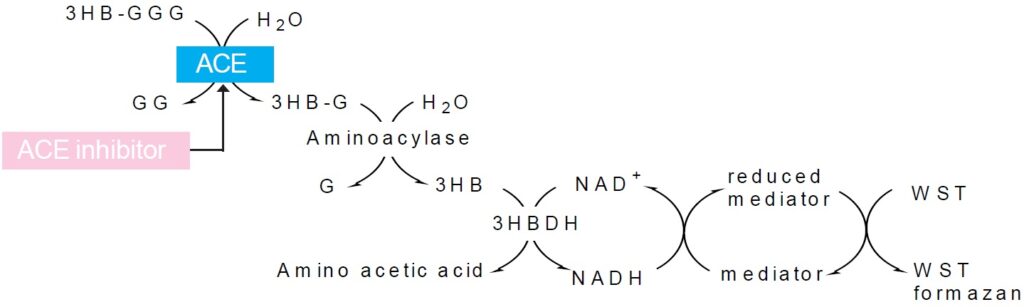
Fig. 1 Principle of the assay system to determine ACE activity or inhibition activity.
Manual
Technical info
plate reader with 450 nm filter; 96-well culture plate, 2-20 μl, 20-200 μl, 100-1000 μl and multi-channel pipettes; 37ºC incubator, Disposable syringe (1 ml)
Required Equipment and Materials

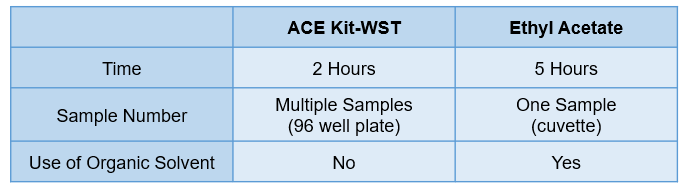
Comparison to the Conventional Method by Ethyl Acetate
Example Assay Data
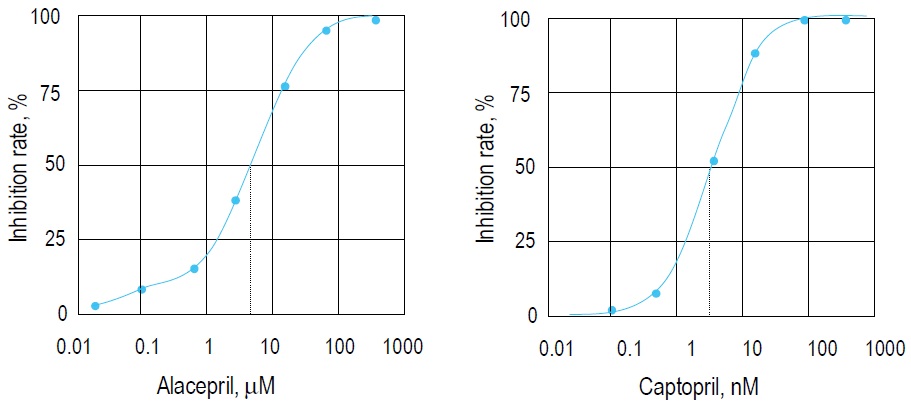
Fig. 2 Inhibition curves prepared by Alacepril and Captopril.
IC50 of Alacepril and Captopril are 3.62 μM and 2.14 nM, respectively. Both compounds are ACE inhibitors.
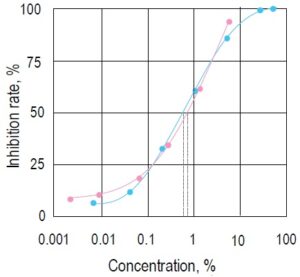
Fig. 3 Inhibition curves prepared by two beverages containing a valyltyrosine (pink●) or lacto tripeptide (blue●) .
IC50 of these beverages are 0.56% and 0.69%, respectively. It is known that these substances have antihypertensive effects. *Concentration of the beverage in the sample solution.
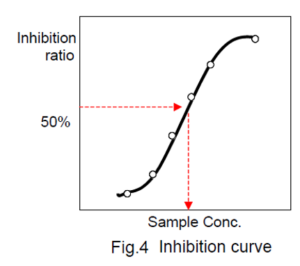
Determination of IC50 (50% inhibitory concentration)
Prepare an inhibit ion curve using the sample concentration for X axis and the ACE inhibitory activity for Y axis. A typical inhibition curve is shown in Fig. 4.
Determine the concentration of the sample solution that gives 50% ACE inhibitory activity as indicated in Fig. 4 .
For 50 tests unit size, 2 samples can be tested in triplicate
Protocol: Determination of IC50
The Potential of ACE Kit-WST in Discovery of Angiotensin-Converting Enzyme (ACE) Inhibitors to Suppress Inflammation after Viral Infection (COVID-19)
ACE Kit-WST is expected to be an advantageous tool to support the discovery of ACE inhibitors effective at suppressing inflammation after SARS-CoV-2 infection.
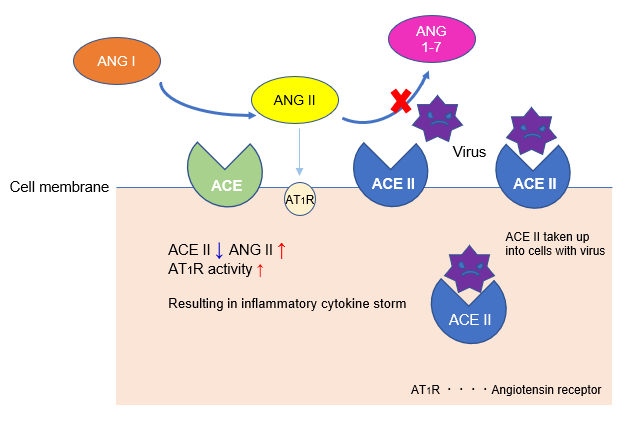
Angiotensin-converting enzyme (ACE) produces angiotensin II (Ang II), a vasoconstrictor, from the C-terminus of angiotensin I (Ang I), which has almost no biological activity. ACE and Ang II activity is countered by ACE2, which functions to cleave inflammatory Ang II into non-inflammatory peptide Ang 1-7.
During SARS-CoV-2 infection, angiotensin-converting enzyme 2 (ACE2) from the cell membrane is taken up into cells along with the virus, decreasing ACE2 activity on the cell membrane. As a result, angiotensin II (Ang II), which had been counteracted by ACE2, becomes uninhibited, beginning a cascade towards an inflammatory cytokine storm that has been suggested to promote lung damage.
Thus, Ang-II mediated cytokine storm is considered to be an important mechanism of severe COVID-19. Therefore, ACE inhibitors, which suppress the Ang II system, are currently expected to reduce the risk of severe Covid-19 symptoms brought on by inflammation, such as acute respiratory distress.
References
1. Hoffmann M, Kleine-Weber H, Schroeder S, et al., “Sars-cov-2 cell entry depends on ace2 and tmprss2 and is blocked by a clinically proven protease inhibitor”, Cell, 2020, 181, 271-280 e278.
2. Imai Y, Kuba K, Rao S, et al., “Angiotensin-converting enzyme 2 protects from severe acute lung failure” Nature, 2005, 436, 112-116.
3. Zou Z, Yan Y, Shu Y, et al., “Angiotensin-converting enzyme 2 protects from lethal avian influenza a h5n1 infections”, Nat Commun., 2014, 5, 3594.
4. Gu H, Xie Z, Li T, et al., “Angiotensin-converting enzyme 2 inhibits lung injury induced by respiratory syncytial virus”, Sci Rep., 2016, 6, 19840.
References
scrollable



Q & A
-
Q
Can ACE activity be measured with this kit?
-
A
ACE activity cannot be measured. This kit can measure ACE inhibitory activity.
-
Q
What are the advantages of this kit?
-
A
Compared to the conventional method (ethyl acetate method), this method is safer, faster, and simpler in the following points.
・No organic solvent is used.
・Multiple samples can be measured at one time.
・Highly reproducible data can be obtained.
-
Q
How many samples can be measured?
-
A
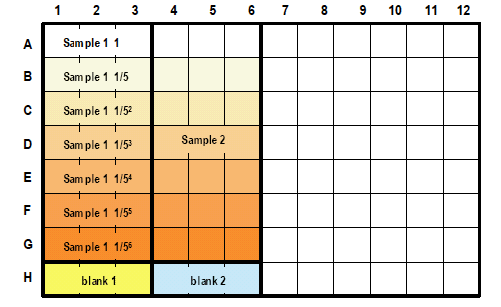
When samples are diluted in 6 steps to obtain IC50 (n=3), 4 samples can be measured in one 96-well microplate.
*To obtain accurate measurement values, use a plate with n=3 or higher.<Figure below> Example of plate layout (n=3)
-
Q
What substances can affect the measurement?
-
A
1. Reducing substances such as ascorbic acid will reduce the WST in Indicator solution and cause a positive error. Please keep the concentration of ascorbic acid at 0.01% or less if the samples contain it.
2. If a substance that inhibits aminoacylase or 3HBDH is present, the enzyme reaction will be affected and accurate measurement will not be possible.
3. Strongly acidic samples containing citric acid or acetic acid may precipitate components in Substrate buffer and inhibit color development. Adjust the sample to pH 5 or higher before measurement.
(Add a small amount of sodium bicarbonate powder to neutralize, centrifuge the sample, and use the supernatant).4) If the sample contains insoluble substances, use the filtered solution or supernatant after centrifugation as the sample.
5) For samples with strong coloration, refer to the instruction manual.
-
Q
The sample I want to measure is difficult to dissolve in water. Is there a way to measure it?
-
A
Samples that are difficult to dissolve in water should be dissolved in DMSO or ethanol and then diluted with water.
We have confirmed that DMSO or ethanol does not interfere with the measurement of organic solvent concentrations up to approximately 1% in the sample.
-
Q
Any examples of extraction methods and precautions?
-
A
Sample Preparation Example (Vegetables/Fruits)
・In the case of food samples, homogenize the sample, centrifuge it, and use the supernatant.
・If the sample is turbid, filter it or dilute it to make a clear solution because the turbidity affects the measurement.
・If the sample has strong coloration, dilute the solution accordingly to the extent that it does not affect the assay. Refer to the instruction manual.
-
Q
Does this kit measure the inhibitory activity against ACE only, or does it react with ACE2 as well?
-
A
ACE only.
This kit does not measure ACE2 inhibitory activity.
-
Q
Is there any problem if I reverse the order of dissolution of Enzyme B and Enzyme A?
-
A
This will reduce sensitivity.
Be sure to dissolve Enzyme B in DI water first, and then use that solution to dissolve Enzyme A.
Handling and storage condition
| 0-5°C |