DTNB

Detection Reagent for SH Moiety
-
Product codeD029 DTNB
-
CAS No.69-78-3
-
Chemical name5,5'-Dithiobis(2-nitrobenzoic acid)
-
MWC14H8N2O8S2=396.35
| Unit size | Price | Item Code |
|---|---|---|
| 1 g | D029-10 |
Description
DTNB is known as Ellman’s reagent. It is used for the colorimetric determination of thiol groups in biological samples. It is fairly soluble in water. Colorless DTNB is converted to yellow 5-Mercapto-2- nitrobenzoic acid in the presence of thiol compounds (Fig. 1). As 5-Mercapto-2-nitrobenzoic acid has an absorption maximum at 412 nm, the absorption spectrum of DTNB does not interfere with thiol detection (Fig. 2).
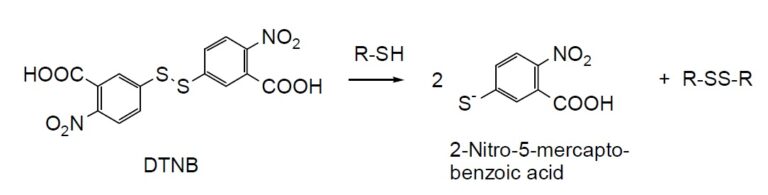
Fig. 1 DTNB reaction with thiol compound
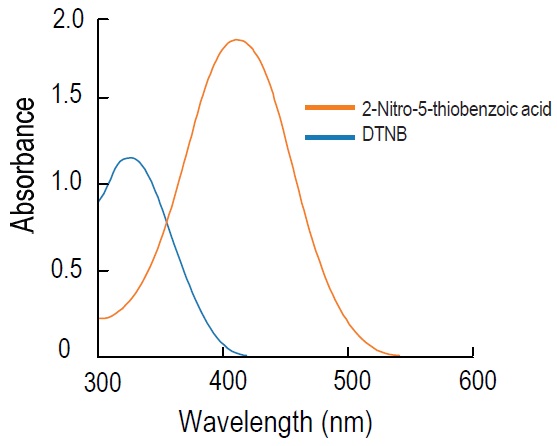
Fig. 2 Absorption spectra of DTNB and reduced DTNB
References
1. D. R. Grassetti, et al., Determination of Sulfhydryl Groups with 2, 2 E or 4, 4 EDithiodipyridine. Arch Biochem Biophys. 1967;119:41-49.
2. R. E. Humphrey, et al., Spectrophotometric Determination of Sulfite with 4, 4 EDithiodipyridine and 5, 5 EDithiobis-(2-nitrobenzoic acid). Anal Chem. 1970;42:698-702.
3. R. E. Humphrey, et al., Spectrophotometric Determination of Cyanide with Organic Disulphides. Talanta. 1971;18:491-497.
4. T. Uete, et al., Spectrophotometric Micromethod for Measuring Cholinesterase Activity in Serum or Plasma. Clin Chem. 1972;18:454-458.
Handling and storage condition
| Appearance: | Pale yellow crystalline powder |
|---|---|
| Solubility in ethyl alcohol: | To pass test (clear, pale yellow) |
| Absorbance: | ≦ 0.195 (420 nm) |
| Solubility in Phosphate buffer: | To pass test (clear, pale yellow) |
| Molar absorptivity: | ≧ 12,000 (around 305 nm) |
| m.p.: | 237 - 247oC (dec.) |
| Loss on drying (105°C): | ≦ 1.0 % |
| Sulfated ash: | ≦ 0.10 % |
| IR spectrum: | Authentic |
| Ambient temperature |











