R-Phycoerythrin Labeling Kit - SH

Antibody / Protein Labeling
- Only 3 hours to get conjugates
- All processes in a single filtration tube
- High recovery of conjugates
- Applicable for 50-200μg IgG
-
Product codeLK26 R-Phycoerythrin Labeling Kit - SH
| Unit size | Price | Item Code |
|---|---|---|
| 3 samples | LK26-10 |
| 3 samples | ・SH-Reactive R-Phycoerythrin ・Reducing Agent ・WS Buffer ・RA Solution ・Reaction Buffer ・Filtration Tube |
3 tubes 3 tubes 4 ml x 1 1 ml x 1 200μlx 1 3 tubes |
|---|
Description
Phycobiliproteins are fluorescent proteins derived from cyanobacteria and eukaryotic algae. Their fluorescence is much higher than chemical fluorescent probes such as fluorescein and rhodamine. R-phycoerythrin (R-PE) is one of the phycobiliproteins and has an orange fluorescence at around 578 nm, and it can be excited at 488 nm (Fig. 1). Because of this high fluorescence, phycobiliprotein-labeled antibodies or other molecules can give greater sensitivity in flow cytometry and immunostaining. R-phycoerythrin Labeling Kit-SH is for simple and rapid preparation of R-PE-labeled IgG (Fig. 2). SH-reactive R-PE (a component of this kit) has a maleimide group and can easily make a covalent bond with a sulfhydryl group of the target molecule without any activation process. The filtration tube in this kit allows a quick buffer exchange and concentration of sample IgG solution. This kit contains all the reagents necessary for R-PE labeling, including the reducing agent for preparation of reduced IgG that has an SH group and the storage buffer for conjugates.
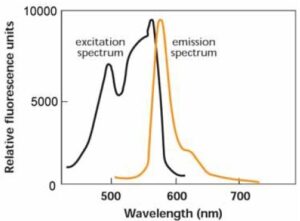
Fig. 1 Fluorescence spectrum of R-PE
Excitation wavelength: 566 nm
Emission wavelength: 578 nm
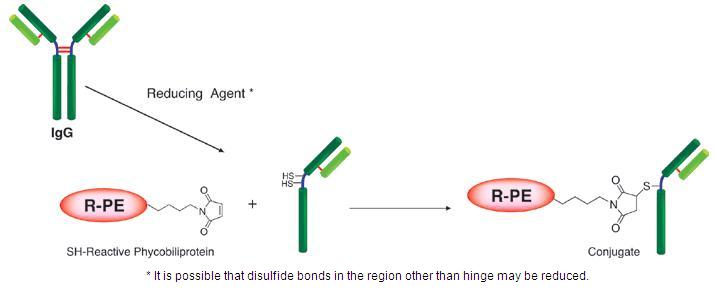
Fig. 2 IgG labeling reaction of SH-reactive R-PE
Precaution
♦ The molecular weight of the reduced protein to be labeled with this kit should be greater than 50,000.
♦ IgG or R-Phycoerythrin-conjugated IgG is always on the membrane of the filtration tube during the labeling process.
♦ If the IgG solution contains other proteins with molecular weight larger than 10,000, such as BSA or gelatin, purify the IgG solution prior to labeling R-phycoerythrin with this kit.
♦ If the IgG solution contains small insoluble materials, centrifuge the solution and use the supernatant for the labeling.
| Developer | Dojindo Molecular Technologies, Inc. |
|---|
Manual
References
1) M. Hikida, S. Casola, N. Takahashi, T. Kaji, T. Takemori, K. Rajewsky and T. Kurosaki, "PLC-γ2 is essential for formation and maintenance of memory B cells", J. Exp. Med.., 2009, 206, (3), 681.
2) M. Segawa, S. Fukada, Y. Yamamoto, H. Yahagi, M. Kanematsu, M. Sato, T. Ito, A. Uezumi, S. Hayashi, Y. Miyagoe-Suzuki, S. Takeda, K. Tsujikawa and H. Yamamoto, "Suppression of macrophage functions impairs skeletal muscle regeneration with severe fibrosis", Exp. Cell Res.., 2008, 314, (17), 3232.
3) M. Yasunaga, S. Saijou, S. Hanaoka, T. Anzai, R. Tsumura, Y. Matsumura, "Significant antitumor effect of an antibody against TMEM180, a new colorectal cancer‐specific molecule", Cancer Sci.., 2019, 110, (2), 761.
Q & A
-
Q
Can I use this kit for F(ab E2?
-
A
Yes, please follow the labeling protocol for IgG. The recovery of the conjugate should be over 80%.
-
Q
Can I use this kit for other proteins?
-
A
Yes, if the molecular weight of the reduced form is greater than 50,000 and it has a reactive SH group, or a disulfide group that can be reduced without losing activity. Follow the labeling protocol for IgG, and use 0.5-1 nmol of sample protein.
-
Q
How many R-PE molecules per reduced IgG are introduced?
-
A
The average number of R-PE molecule per reduced IgG is 1 to 2.
-
Q
Do I have to use the Filtration tube prior to labeling the protein?
-
A
If the protein solution does not contain small molecules with reactive SH groups and the concentration of the protein is 10 mg per ml, or about 70 μM, there is no need to use the Filtration tube. Just mix 10 μl of the sample solution with Reaction buffer and add the mixture to a vial of the SH-reactive R-PE.
-
Q
Do I have to use WS buffer included with the kit?
-
A
Yes. Use the WS buffer to prepare a stock solution of the conjugate. However, you can choose any kind of buffer appropriate to dilute the conjugate stock solution for your experiment.
Handling and storage condition
| 0-5°C, Protect from moisture | |
|
Danger / harmful symbol mark |

|
|---|---|











