ICG Labeling Kit - NH2

Antibody / Protein Labeling
- Suitable wavelength for In Vivo imaging
- Quick and Easy Labeling to Antibodies : 1.5hr
- High Recovery Rate : more than 90%
-
Product codeLK31 ICG Labeling Kit - NH2
| Unit size | Price | Item Code |
|---|---|---|
| 3 samples | LK31-10 |
| 3 samples | ・NH2-Reactive ICG ・WS Buffer ・Reaction Buffer ・Filtration Tube |
3 tubes 4 ml x1 500 μlx1 3 tubes |
|---|
Description
ICG Labeling Kit – NH2 is used primarily for the preparation of ICG (Iindocyanin green)-labeled antibody for near-infrared fluorescence imaging. ICG offers two remarkable properties:
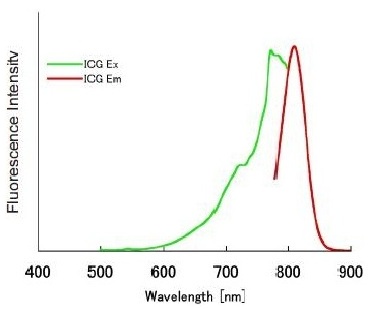
1) ICG has a strong near-infrared fluorescence even after a few days under physiological conditions. The excitation and emission wavelength of the ICG-labeled proteins are 774 nm and 805 nm, respectively.
2) ICG has been used in clinical fields such as a hepatic deficiency testing. Therefore, ICG and ICG conjugates are materials suitable for in vivo imaging.
This kit contains all required compornents required for labeling, including storage buffer for conjugates. The labeling process is simple:. Add NH2-reactive ICG to protein solution on a filter membrane, and incubate at 37ºC, for 10 minutes. A filtration tube can remove excess ICG molecules.
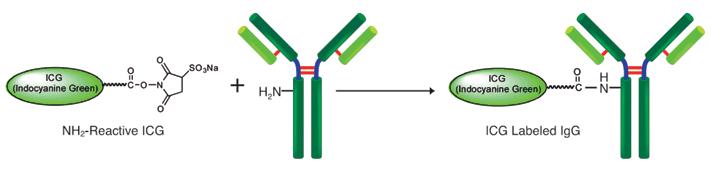
Fig. 1 Fluorescein Labeling Process to IgG
Precaution
♦ If the IgG solution contains other proteins with molecular weight greater than 10,000, such as serum albumin or gelatin, purify the IgG solution before labeling fluorescein with this kit. Commercially available antibody may contain BSA or gelatin as a stabilizer.
| Developer | Dojindo Molecular Technologies, Inc. |
|---|
Manual
Technical info
-
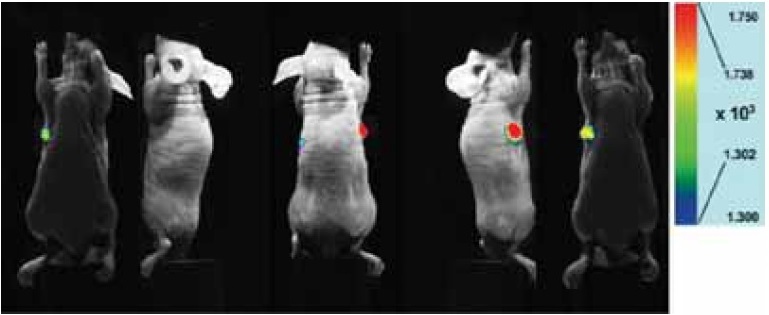
Fig. 2 Fluorescent Overlay Imaging
-
A mouse subcutaneous tumor model at 48 hours post injection of ICG labeled antibody(50 μg) shows clear evidence of selective accumulation in the tumor.
mouse:
BALB/c nu nu (female, 11 weeks)
tumor cell:
HeLa (4 weeks after transplant in subcutaneous right axilla)
antibody:
anti-integrin a2 antibody
measurement condition:
Ex 785 nm, Em 845/55 nm/center/band), exposure time: 10 sec
References
1) M. Ogawa, C. A. S. Regino, J. Seidel, M. V. Green, W. Xi, M. Williams, N. Kosaka, P. L. Choyke and H. Kobayashi, "Dual-Modality Molecular Imaging Using Antibodies Labeled with Activatable Fluorescence and a Radionuclide for Specific and Quantitative Targeted Cancer Detection", Bioconjugate Chem., 2009, 20(11), 2177.
2) M. Ogawa, N. Kosaka, P. L. Choyke and H. Kobayashi, "In vivo Molecular Imaging of Cancer with a Quenching Near-Infrared Fluorescent Probe Using Conjuates of Monoclonal Antibodies and Indocyanine Green", Cancer Res., 2009, 69(4), 1268.
3) N. Kosaka, M. Ogawa, P. L. Choyke and H. Kobayashi, "Clinical implications of near-infrared fluorescence imaging in cancer", Future Oncology, 2009, 5(9), 1501.
4) S. Ito, N.Muguruma, S. Hayashi, S. Taoka, T. Bando, K. Inayama, M. Sogabe, T. Okahisa, S. Okamura, H. Shibata, T. Irimura, K. Takesako and S. Shibamura, "Development of Agents for Reinforcement of Fluorescence on Near-infrared Ray Excitation for Immunohistological Staining", Bioorg. Med. Chem., 1998, 6, 613.
4) S. Ito, N. Muguruma, Y. Kakehashi, S. Hayashi, S. Okamura, H. Shibata, T. Okahisa, M. Kanamori, S. Shibamura, K. Takesako, M. Nozawa, K. Ishida and M. Shiga, "Development of Fluorescence-Emitting Antibody Labeling Substance by Near-Infrared Ray Excitation", Bioorg. Med. Chem. Lett., 1995, 5, 2689.
6) K. Inayama, S. Ito, N. Muguruma, Y. Kusaka, T. Bando, Y. Tadatsu, M. Tadatsu, K. Ii, S. Shibamura and K. Takesako, "Basic Study of an Agent for Reinforcement of Near-infrared Fluorescence on Tumor Tissue", Digestive and Liver Disease, 2003, 35, 88.
7) S. Ito, N. Muguruma, S. Hayashi, S. Taoka, T. Bando, Y. Kusaka, M. Yano, S. Ichikawa, A. Hiasa, T. Omoya, H. Honda, I. Shimizu, K. Ii, K. Nakamura, K. Takesako, Y. Goto and S. Shibamura, "Visualization of Human Gastric Cancer with a Novel Infrared Fluorescent Labeling Marker of Anti-carcinoembryonic Antigen Antibody in vitro", Dig. Endosc., 2000, 12, 33.
8) S. Taoka, S. Ito, N. Muguruma, S. Hayashi, Y. Kusaka, K. Ii, K. Nakamura, K. Imaizumi, K. Takesako and S. Shibamura, "Reflected Illumination-type Imaging System for the Development of Infrared Fluorescence Endoscopy", Dig. Endosc.,1999, 11(4), 321.
9) S. Ito, N. Muguruma, S. Hayashi, S. Taoka, A. Tsutsui, T. Fukuda, T. Okahisa, Y. Ohkita, H. Matsunaga, I. Shimizu, K. Nakamura, K. Imaizumi, K. Takesako and S. Shibamura,"Development of an Imaging System Using Fluorescent Labeling Substances Excited by Infrared Rays", Dig. Endosc., 1997, 9, 278.
10) S. Ito, N. Muguruma, Y. Kusaka, M. Tadatsu, K. Inayama, Y. Musashi, M. Yano, T. Bando, H. Honda, I. Shimizu, K. Ii, K. Takesako, H. Takeuchi and S. Shibamura, "Detection of Human Ganstric Cancer of Resected Specimens Using a Novel Infrared Fluorescent Anti-Human Carcinoembryonic Antigen Antibody with an Infrared Fluorescence Endoscope in Vitro", Endoscopy, 2001, 33(10), 849.
11) N. Muguruma, S. Ito, T. Bando, S. Taoka, Y. Kusaka, S. Hayashi, S. Ichikawa, Y. Matsunaga, Y. Tada, S. Okamura, K. Ii, K. Imaizumi, K. Nakamura, K. Takesako and S. Shibamura, "Labeled Carcinoembryonic Antigen Antibodies Excitable by Infrared Rays: a Novel Diagnostic Method for Micro Cancers in the Digestive Tract", Internal Medicine, 1999, 38(7), 537.
12) T. Bando, N. Muguruma, S. Ito, Y. Musashi, K. Inayama, Y. Kusaka, M. Tadatsu, K. Ii, T. Irimura, S. Shibamura and K. Takesako, "Basic Study on a Labeled anti-mucin Antibody Detectable by Infrared-fluorescence Endoscopy", J. Gastroenterol., 2002, 37, 260.
13) N. Muguruma, S. Ito, S. Hayashi, S. Taoka, H. Kakehashi, K. Ii, S. Shibamura and K. Takesako, "Antibodies Labeled with Fluorescence-agent Excitable by Infrared Rays", J. Gastroenterol., 1998, 33, 467.
14) W. Aung, A. Tsuji, H. Sudo, A. Sugyo, T. Furukawa, Y. Ukai, Y. Kurosawa and T. Saga, "Immunotargeting of Integrin α6β4 for Single-Photon Emission Computed Tomography and Near-Infrared Fluorescence Imaging in a Pancreatic Cancer Model", Molecular Imaging, 2016, 15, 1.
Handling and storage condition
| 0-5°C, Protect from moisture |











