Biotin-AC5 Sulfo-OSu

Biotin Labeling Reagent
-
Product codeB320 Biotin-AC5 Sulfo-OSu
-
CAS No.109940-19-4(free acid)
-
Chemical name6-(Biotinylamino)hexanoic acid N-hydroxysulfosuccinimide ester
-
MWC20H29N4NaO9S2=556.59
| Unit size | Price | Item Code |
|---|---|---|
| 10 mg | B320-10 |
Description
Product Description of Amine-Reactive Biotins
The avidin-biotin system has many applications in immunology and histochemistry. The interaction between avidin and biotin is remarkably strong with a dissociation constant on the order of 10-15 M. Biotin is usually added to primary or secondary antibodies such as anti-IgG and anti-IgM. After preparing the antigen-antibody complex with the biotin-labeled antibody, colorimetric, or fluorometric detection of the antigen is performed using enzyme or fluorescein-labeled avidin or streptavidin. Succinimidyl ester biotins react with primary and secondary amines, such as amino acids and proteins, at pH 7-9. Succinimidyl ester reacts with free amine groups to create a stable amide bond. Succinimidyl biotin reagents must be dissolved in DMSO, DMF, or alcohol. Stock solutions prepared with DMSO are stable for several months at -20ºC. Sulfo succinimidyl biotin reagents are soluble in water, so there is no need to use organic solvents such as DMF or DMSO. IgG prepared using biotin with a longer spacer such as Biotin-(AC5)2-OSu or Biotin-(AC5)2-Sulfo-OSu, has a better signal-to-noise ratio. The longer spacer enables streptavidin or anti-biotin IgG to recognize biotin without structural inhibition. Therefore, Biotin-(AC5)2-OSu is utilized as the biotin labeling agent in the Biotin Labeling Kit-NH2.
Reaction Scheme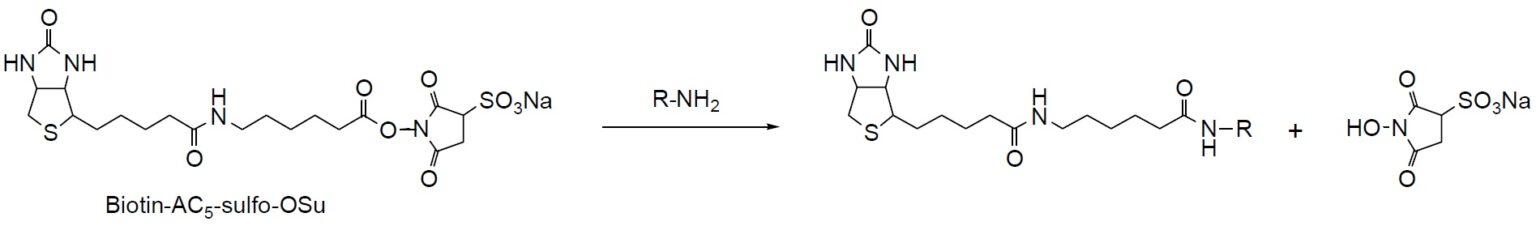
Technical info
Labeling Procedure for IgG
1. Prepare 10 mM of the biotin labeling reagent using DMSO.
2. Prepare 100 μl of 1 mg per ml IgG buffer solution (pH 7.5-8.5) that does not contain any large molecules with amine compounds.
3. Add 1-5ml biotin labeling reagent DMSO solution to the IgG buffer solution and incubate at 37ºC for 1 hour.
4. Remove excess biotin labeling reagent using gel filtration or dialysis.
5. Prepare solutions for further experiment using an appropriate buffer such as PBST (0.05% Tween 20/PBS).
References
1) J. Wormmeester, F. Stiekema and C. Groot, "Immunoselective Cell Separation", Methods Enzymol., 1990, 184, 314.
2) J. J. Leary, D. J. Brigati and D. C. Ward, "Rapid and Sensitive Colorimetric Method for Visualizing Biotin-labeled DNA Probes Hybridized to DNA or RNA Immobilized on Nitrocellulose: Bio-blots", Proc. Batl. Acad. Sci. USA, 1983, 80, 4045.
3) W. T. Lee and D. H. Conrad, "The Murine Lymphocyte Receptor for IgE. II. Characterization of the Multivalent Nature of the B Lymphocyte Receptor for IgE", J. Exp. Med., 1984, 159, 1790.
4) D. R. Gretch, M. Suter and M. F. Stinski, "The use of Biotinylated Monoclonal Antibodies and Streptavidin Affinity Chromatography to Isolate Herpesvirus Hydrophobic Proteins or Glycoproteins", Anal. Biochem., 1987, 163, 270.
5) M. Shimkus, J. Levy and T.Herman, "A Chemically Cleavable Biotinylated Nucleotide: Usefulness in the Recovery of Protein-DNA Complexes from Avidin Affinity Columns", Proc. Natl. Acad. Sci. USA, 1985, 82, 2593.
6) W. J. LaRochelle and S. C. Froehner, "Immunochemical Detection of Proteins Biotinylated on Nitrocellulose Replicas", J. Immunol. Methods, 1986, 92, 65.
7) P. S. Anjaneyulu and J. V. Staros, "Reactions of N-hydroxysulfosuccinimide Active Esters", Int. J. Pep. Protein Res., 1987, 30, 117.
8) H. M. Ingalls, C. M. Goodloe-Holland and E. J. Luna, "Junctional Plasma Membrane Domains Isolated from Aggregating Dictyostelium Discoideum Amebae", Proc. Natl. Acad. Sci. USA, 1986, 83, 4779.
9) J. L. Guesdon, T. Ternynck and S. Avrameas, "The Use of Avidin-biotin Interaction in Immunoenzymatic Techniques", J. Histochem, Cytochem, 1979, 27, 1131.
Handling and storage condition
| Appearance: | White to pale reddish brown powder |
|---|---|
| Purity (HPLC): | ≧ 95.0 % |
| Solubility in water: | To pass test (clear, colorless to slightly brown) |
| NMR spectrum: | Authentic |
| -20°C |








