-SulfoBiotics- SSP4

Biosulfur Analysis
-
Product codeSB10 -SulfoBiotics- SSP4
-
CAS No.1810731-98-6
-
Chemical name3',6'-Di(O-thiosalicyl)fluorescein
-
MWC34H20O7S2=604.65
| Unit size | Price | Item Code |
|---|---|---|
| 1 mg | SB10-10 |
Product Description
It becomes obvious that there are a lots of molecules containing sulfane sulfurs such as persulfides and polysulfides in living organism. These molecular species are involved in production, storage and release of hydrogen sulfide, which is recognized as an important physiological mediator. Furthermore, recent studies reveal that persulfides and polysulfides may control intracellular signal transduction through s-sulfhydration of proteins.
SSP4 (Sulfane Sulfur Probe 4) is a novel fluorescent probe to detect sulfane sulfurs selectively. SSP4 itself is non-fluorescent, but it emits strong green fluorescence when it reacts with sulfane sulfurs. Thus, SSP4 enables high sensitive fluorescence detection and imaging of sulfane sulfurs.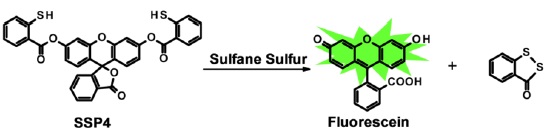
Fig. 1 Chemical Reaction with sulfane sulfer
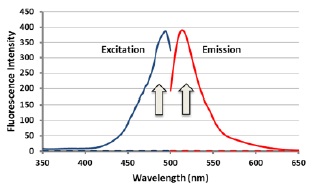
Fig. 2 Fluorescence spectrum (Em: 482nm/Ex: 515nm)
Chemical species containing sulfane sulfurs

Fig. 3 The formation of sulfane sulfer in vivo changes due to reduction/oxidation and translation
| Developer | Dojindo Molecular Technologies, Inc. |
|---|
Manual
Technical info
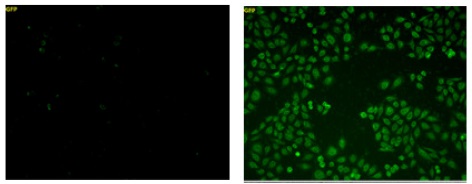
Left: Control cells
Right: Treatment of 100 μmol/l Na2S3
Cell: CHO cell
Exposure time: 1,000 msc
References
1) W. Chen, C. Liu, B. Peng, Y. Zhao, A. Pacheco, and M. Xian, “New fluorescent probe for sulfane sulfurs and the application in bioimaging”, Chem. Sci., 2013, 4, 2892.
2) T. Ida, T. Sawa, H. Ihara. Y. Tsuchiya, Y. Watanabe, Y. Kumagai, M. Suematsu, H. Motohashi, S. Fujii, T, Matsunaga, M. Yamamoto, K. Ono, N. O. Devarie-Baez, M. Xian, J. M Fukuto, and T. Akaike, “Reactive cysteine persulfides and S-polythiolation regulate oxidative stress and redox signaling”, Proc Natl Acad Sci U S A., 2014, 111, 7606.
3) E. Marutani, M. Sakaguchi, W. Chen, K. Sasakura, J. Liu, M. Xian, K. Hanaoka, T. Nagano, and F. Ichinose, "Cytoprotective effects of hydrogen sulfide-releasing N-methyl-D-aspartate receptor antagonists mediated by intracellular sulfane sulfur", Med. Chem. Commun., 2014, 5, 1577.
4) M. Sakaguchi, E. Marutani, H-S. Shin, W. Chen, K. Hanaoka, M. Xian, and F. Ichinose, “Sodium Thiosulfate Attenuates Acute Lung Injury in Mice”, Anesthesiology, 2014, 121, 1248
5) Y. Kimura, Y. Toyofuku, S. Koike, N. Shibuya, N. Nagahara, D. Lefer, Y. Ogasawara, and H. Kimura, "Identification of H2S3 and H2S produced by 3-mercaptopyruvate sulfurtransferase in the brain", Nature Scientific Reports., 2015, 5, 14774.
6) E. DeLeon, Y. Gao, E. Huang, M. Arif, N. Arora, A. Divietro, S. Patel, K. Olson, "A case of mistaken identity: are reactive oxygen species actually reactive sulfide species?", Am J Physiol Regul Integr Comp Physiol, 2016, 310(7), R549.
7) P. Yadav, M. Martinov, V. Vitvitsky, J. Seravalli, R. Wedmann, M. Filipovic, and R. Banerjee, "Biosynthesis and Reactivity of Cysteine Persulfides in Signaling", Jounal of the American Chemical Society., 2016, 138, (1), 289.
8)A. Moustafa, Y. Habara, "Reciprocal interaction among gasotransmitters in isolated pancreatic β-cells", Free Radical Biology and Medicine., 2016, 90, 47.
9) A. Moustafa and Y. Habara, "Crosstalk between polysulfide and nitric oxide in rat peritoneal mast cells", Am. J. Physiol. Cell Physiol., (in press)
10) E. R. DeLeon, Y. Gao, E. Huang, M. Arif, N. Arora, A. Divietro, S. Patel, and K. R. Olson , "A case of mistaken identity: are reactive oxygen species actually reactive sulfide species?", American Journal of Physiology: Regulatory, Integrative and Comparative Physiology ., 2015, 310, (7), 549.
11) N. Takahashi, FY Wei, S. Watanabe, M. Hirayama, Y. Ohuchi, A. Fujimura, T. Kaitsuka, I. Ishii, T. Sawa, H. Nakayama, T. Akaike and K. Tomizawa, "Reactive sulfur species regulate tRNA methylthiolation and contribute to insulin secretion", Nucleic Acids Res., 2016, doi: 10.1093/nar/gkw745 .
12) D. Garai, BB. R. Gonzalez, PG. Furtmuller, JM. Fukuto, M. Xian, J. L. Garriga, C. Obinger and P. Nagy
, "Mechanisms of myeloperoxidase catalyzed oxidation of H2S by H2O2 or O2 to produce potent protein Cys-polysulfide-inducing species.", Free Radic. Biol. Med.., 2017, 113, 551.
13) Ningke Hou, Zhenzhen Yan, Kaili Fan, Huanjie Li, Rui Zhao, Yongzhen Xia, Luying Xun, Huaiwei Liu , 'OxyR senses sulfane sulfur and activates the genes for its removal in Escherichia coli', Redox Biology., 2019,doi.org/10.1016/j.redox.2019.101293 .
Handling and storage condition
| Appearance: | White to pale yellow powder or solid |
|---|---|
| Purity (HPLC): | ≧ 90.0 % |
| -20°C, Protect from light |












