-SulfoBiotics- GYY4137

Biosulfur Analysis
-
Product codeSB06 -SulfoBiotics- GYY4137
-
CAS No.106740-09-4
-
Chemical name(4-Methoxyphenyl)morpholinylphosphinodithioic acid, morpholine salt
-
MWC15H25N2O3PS2=376.47
| Unit size | Price | Item Code |
|---|---|---|
| 10 mg | SB06-10 |
Product Description
GYY4137 is one of the synthetic hydrogen sulfide donors derived from Lawsson’s reagent. It is water-soluble and releases H2S very slowly by hydrolysis in aqueous solutions. GYY4137 has shown distinct cellular effects such as anti-hypertensive, anti-atherosclerotic and anti-tumor activities in several reports.
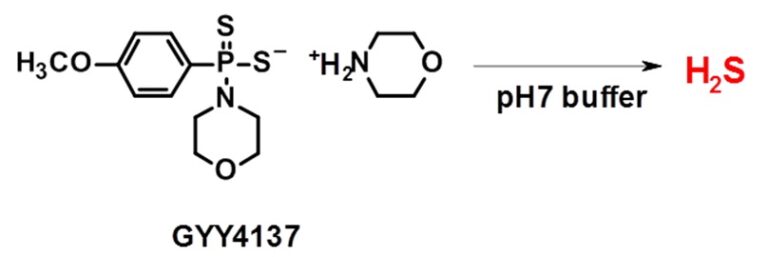
Fig. 1 H2S Releasing Mechanism of GYY4137
| Developer | Dojindo Molecular Technologies, Inc. |
|---|
Manual
Technical info
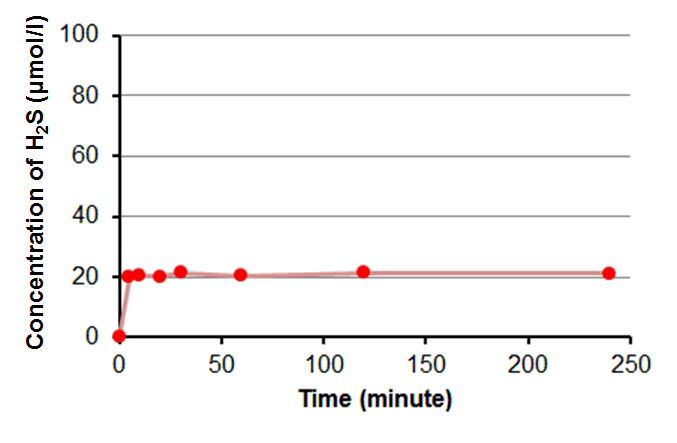
H2S Releasing Curve of GYY4137 in PBS (100 μM)
References
1) L. Li, M. Whiteman, Y. Y. Guan, K. L. Neo, Y. Cheng, S. W. Lee, Y. Zhao, R. Baskar, C-H. Tan, and P. K. Moore, “Characterization of a Novel, Water-Soluble Hydrogen Sulfide-Releasing Molecule (GYY4137): New Insights Into the Biology of Hydrogen Sulfide”, Circulation, 2008, 117, 2351.
2) M. Whiteman, L. Li, P. Rose, C-H. Tan, D. B. Parkinson, and P. K. Moore, “The Effect of Hydrogen Sulfide Donors of Lipopolysaccharide-Induced Formation of Inflammatory Mediators in Macrophages”, Antioxid. Redox Signal., 2013, 19, 1749.
3) Z. W. Lee, J. Zhou, C-S. Chen, Y. Zhao, C-H. Tan, L. Li, P. K. Moore, and L-W. Deng, “The Slow-Releasing Hydrogen Sulfide Donor, GYY4137, Exhibits Novel Anti-Cancer Effects In Vitro and In Vivo”, PLos One, 2011, 6, e21077.
4) L. Li, B. Fox, J. Keeble, M. Salto-Tellez, P. G. Winyard, M. E. Wood, P. K. Moore, and M. Whiteman, “The complex effects of the slow-releasing hydrogen sulfide donor GYY4137 in a model of acute joint inflammation and in human cartilage cells”, J. Cell. Mol. Med., 2013, 17, 365.
5) Z. Liu, Y. Han, L. Li, G. Meng, X. Li, M. Shirhan, M. T. Peh, L. Xie, S. Zhou, X. Wang, Q. Chen, W. Dai, C-H. Tan, S. Pan, P. K. Moore and Y. Ji, “The hydrogen sulfide donor, GYY4137, exhibits anti-atherosclerotic activity in high fat fed apolipoprotein E-/- mice”, Br. J. Pharmacology, 2013, 169, 1795.
6) Satoru Sakuma, Saaya Minamino, MayaTakase, Yoshitaka Ishiyama, Hiroyuki Hosokura, Tetsuya Kohda, Yukino Ikeda, Yohko Fujimoto, "Hydrogen sulfide donor GYY4137 suppresses proliferation of human colorectal cancer Caco-2 cells by inducing both cell cycle arrest and cell death", Heliyon., 2019,doi.org/10.1016/j.heliyon.2019.e02244 .
Handling and storage condition
| Appearance: | White to slightly yellow powder |
|---|---|
| Purity (HPLC): | ≧ 95.0 % |
| NMR spectrum: | Authentic |
| 0-5°C | |
|
Danger / harmful symbol mark |
 
|
|---|---|