-SulfoBiotics- PEG-PCMal

Biosulfur Analysis
-
Product codeSB20 -SulfoBiotics- PEG-PCMal
-
MW2736.19
| Unit size | Price | Item Code |
|---|---|---|
| 1 mg | SB20-01 |
Description
Modification of protein thiols is one of the most important post-translational modifications and it occurs according to the redox states in cells. It has recently been revealed that the modifications of thiol groups control cellular functions such as transcription, protein expression, cell death, etc. Therefore, detection of the redox states of the individual protein is important to understand cellular events. -SulfoBiotics- PEG-PCMal is a reagent to visualize the redox states of protein by electrophoretic analysis. PEG-PCMal has a maleimide group that can bind covalently to a protein thiol group. A mobility shift corresponding to about 5 kDa is observed by the electrophoretic analysis when one molecule of PEG-PCMal binds to a thiol group of the target protein. Thus, the number of free thiol groups on a protein can be clearly identified by SDS-PAGE through the mobility shift assay. The conventional reagents, PEG-Maleimide, have been widely used in mobility shift assays, however, transfer efficiency and antibody recognition on western blot are low due to the PEG chains labeled in protein. Because this reagent has a UV photocleavable moiety in the molecule, the PEG chains are cut off from the labeled protein in the gel with UV irradiation after the electrophoresis. Therefore, the protein treated with UV irradiation can be transferred from the gel to PVDF membrane and detected by antibodies.
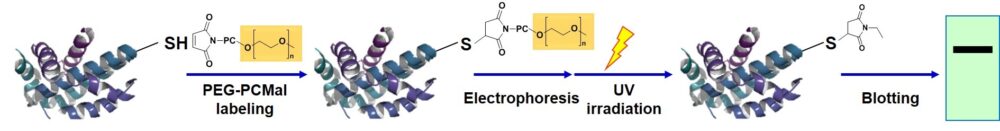
Figure 1 Schematic protocol of PEG-PCMal
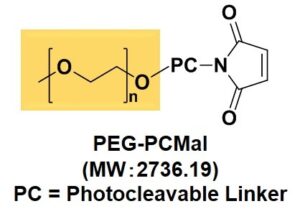
Figure 2 Structure of PEG-PCMal
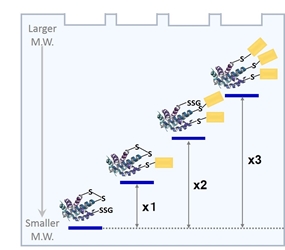
Figure 3 Gel shift assay mechanism by PEG-PCMal depend on protein redox state
| Developer | Dojindo Molecular Technologies, Inc. |
|---|
Manual
Technical info
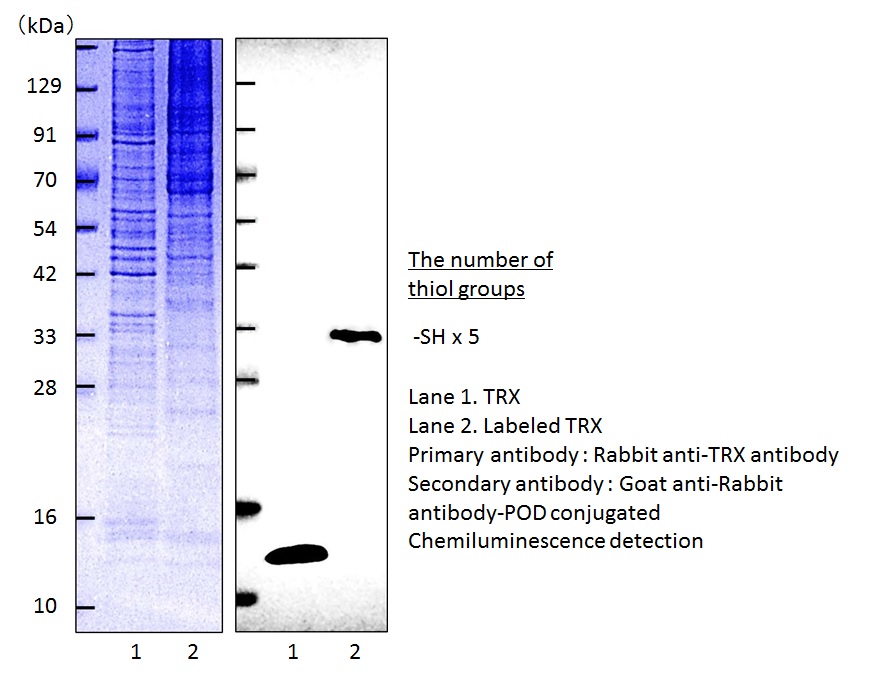
Figure 4 Visualization of the redox state of the TRX in HeLa cells
Analysis of the redox states of protein (ATP synthase γ subunit) in Arabidopsis thaliana
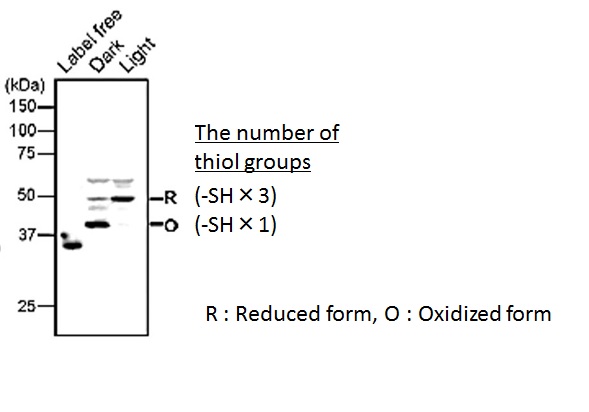
Figure 5 Visualization of the redox state of the photoresponsive protein in Arabidopsis thaliana
Technical advisor
Dr. Toru Hisabori (Laboratory for Chemistry and Life Science, Institute of Innovative Research, Tokyo Institute of Technology)
Dr. Keisuke Yoshida (Laboratory for Chemistry and Life Science, Institute of Innovative Research, Tokyo Institute of Technology)
Dr. Satoshi Hara (School of Life Science and Technology, Tokyo Institute of Technology)
References
1. L. Makmura, M. Hamann, A. Areopagita, S. Furuta, A. Munoz and J. Momand. , “Development of a sensitive assay to detect reversibly oxidized protein cysteine sulfhydryl groups”, Antioxid Redox Signal., 2001, 3, (6), 1105.
2. HH Wu, J.A. Thomas and J. Momand, “p53 protein oxidation in cultured cells in response to pyrrolidine dithiocarbamate: a novel method for relating the amount of p53 oxidation in vivo to the regulation of p53-responsive genes”, Biochem J., 2000, 351, 87.
3. JR. Burgoyne, O. Oviosu and P. Eaton., “The PEG-switch assay: A fast semi-quantitative method to determine protein reversible cysteine oxidation”, J Pharmacol Toxicol Methods., 2013, 68, (3), 297.
4. L. JTetsch, C. Koller, A. Donhofer and K. Jung, “Detection and function of an intramolecular disulfide bond in the pH-responsive CadC of Escherichia coli”, BMC Microbiol., 2011, doi: 10.1186/1471-2180-11-74.
Handling and storage condition
| Appearance: | Slightly yellow to pale yellowish brown powder or solid |
|---|---|
| Sensitivity: | To pass test |
| 0-5°C, Protect from light |









