-SulfoBiotics- HSip-1 DA

Biosulfur Analysis
-
Product codeSB22 -SulfoBiotics- HSip-1 DA
-
CAS No.1346170-03-3(free base)
-
Chemical nameN-[3',6'-Bis(acetyloxy)-3-oxo-3H-spiro(isobenzofuraN-1,9'-xanthen)-5-yl]-(1,4,7,10-tetraazacyclododecane-1-acetamide-κN1,κN4,κN7,κN10copper(2+)bis(trifluoroacetat
-
MWC38H37CuF6N5O12=933.26
| Unit size | Price | Item Code |
|---|---|---|
| 50 µg | SB22-10 |
Description
It has been recognized that hydrogen sulfide (H2S) has an important role as a physiological active substance for vasodilation, cytoprotection, and modulation of insulin secretion. H2S is considered as a gaseous molecule such as nitric oxide and carbon monoxide. However, around 80% of the total sulfide exists as hydrogen sulfide anion (HS- ) under physiological condition, since the pKa is about 7. In addition, HS- easily converts to various biochemical molecules such as persulfides and polysulfides, which react with sulfhydryl moieties in a living body. -SulfoBiotics- HSip-1 is a novel fluorescent probe to detect H2S selectively and it emits strong green fluorescence when it reacts with H2S. -SulfoBiotics-HSip-1 DA is cell membrane permeable and it enables fluorescent imaging of intracellular H2S.
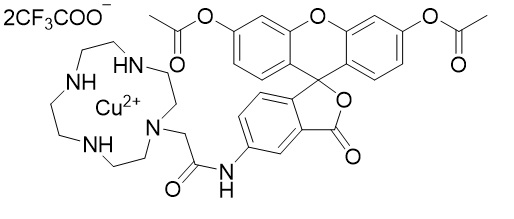
Fig. 1 Chemical structure of HSip-1 DA
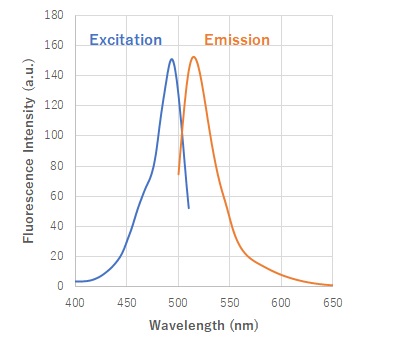
Fig. 2 Excitation and emission spectra of HSip-1 reacted with H2S
(Em: 491nm/Ex: 516nm)
| Developer | Dojindo Molecular Technologies, Inc. |
|---|
Manual
Technical info
1) HeLa cells were seeded on μ-slide 8 well (Ibidi) and cultured at 37℃ overnight in a 5% CO2 incubator.
2) The culture medium was discarded and the cells were washed with a serum-free medium (MEM) twice.
3) HSip-1 DA stock solution (1 mmol/l) was diluted with a serum-free medium (MEM) to prepare 5 μmol/l HSip-1 DA working solution. *Please optimize the final concentration of HSip-1 DA depeneding on the cell lines.
4) HSip-1 DA working solution (5 μmol/l, 200 μl) was added to the cells, and the cells were cultured at 37℃ for 30 minutes in a 5% CO2 incubator.
5) The supernatant was discarded, and the cells were washed with HBSS twice.
6) Na2S solution (200 μmol/l, 200 μl) was added to the each well, and the cells were cultured at 37℃ for 30 minutes in a 5% CO2 incubator.
7) The supernatant was discarded and the cells were washed with HBSS twice.
8) HBSS (200 μl) were added, and the cells were observed by confocal fluorescence microscopy.
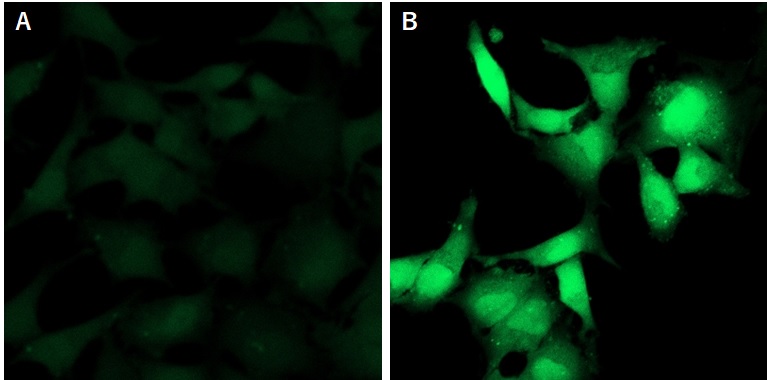
Fig. Detection of hydrogen sulfide using HSip-1 DA in HeLa cells treated with Na2S.
(A: Control, B: 200 μmol/l Na2S treated)
References
1) K.Sasakura, K.Hanaoka, N.Shibuya, Y.Mikami, Y. Kimura, T.Komatsu, T.Ueno, T.Terai, H.Kimura, and Tetsuo Nagano, "Development of a Highly Selective Fluorescence Probe for Hydrogen Sulfide", J. Am. Chem. Soc., 2011, 133, (45), 18003–18005.
2) H.Kimura, N.Shibuya, and Y.Kimura, "Hydrogen Sulfide Is a Signaling Molecule and a Cytoprotectant", Antioxid Redox Signal., 2012, 17, (1), 45-47.
3) E.Marutani, M. Sakaguchi, W.Chen, K.Sasakura,c J. Liu, M.Xian, K. Hanaoka, T.Nagano, and F.Ichinose, "Cytoprotective effects of hydrogen sulfide-releasing N-methyl-D-aspartate receptor antagonists are mediated by intracellular sulfane sulfur", Medchemcomm., 2014, 5, (10), 1577-1583.
4) N.Fukushima, N.Ieda, M.Kawaguchi, K. Sasakura, T.Nagano, K.Hanaoka, N.Miyata, H.Nakagawa, "Development of photo-controllable hydrogen sulfide donor applicable in live cells", Bioorganic & Medicinal Chemistry Letters., 2015, 25, (2), 175-178.
5) E.Marutani, M.Yamada, T.Ida, K.Tokuda, K.Ikeda, S.Kai, K.Shirozu, K.Hayashida, S.Kosugi, K.Hanaoka, M.Kaneki, T.Akaike, and F.Ichinose, "Thiosulfate Mediates Cytoprotective Effects of Hydrogen Sulfide Against Neuronal Ischemia", J Am Heart Assoc., 2015, 4, (11), e002125.
Handling and storage condition
| Appearance: | Blue solid |
|---|---|
| Purity (HPLC): | ≧ 90.0 % |
| Fluorescence spectrum: | To pass test |
| -20°C, Nitrogen substitution |












