AB-NTA free acid

Chelate Labeling Reagent
-
Product codeA459 AB-NTA free acid
-
CAS No.129179-17-5
-
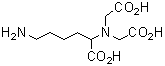
Chemical nameN-(5-Amino-1-carboxypentyl)iminodiacetic acid
-
MWC10H18N2O6=262.26
| Unit size | Price | Item Code |
|---|---|---|
| 100 mg | A459-10 |
Description
Product Description
Aminobutyl-NTA (AB-NTA) free acid replaces AB-NTA (disodium salt form; former Product Code: A296-10). The new free acid form is less hygroscopic than the disodium salt form. Dissolving the new acid form in a buffer involves the same procedure as dissolving disodium salt in a buffer. The new acid form can also be dissolved in water with sonication (up to 3%). Aminobutyl-NTA was first used by Dr. Hochuli in 1987 to purify recombinant proteins (the “Histag Etechnique). Since then, the AB=NTA has become an indispensable tool for immobilizing proteins with high specificity on solid surfaces, such as glass or the gold electrode. A solid surface is modified by AB-NTA and bio-functionalized via Ni (II) with a genetically expressed protein bearing a hexahistidine extension at its terminus. The His-tag technique has become increasingly important, particularl in surface plasmon resonance and structural analyses of proteins by x-ray interference. Using the His-tag technique, Dr. Noji was able to directly observe the rotation of F1-ATPase by fluorescence microscope.
Chemical Structure
References
1. E. Hochuli, H. Doeli and A. Schacher, New Metal Chelate Adsorbent Selective for Proteins and Peptides Containing Neighbouring Histidine Residues, J. Chromatogr., 1987, 411, 177.
2. E. Hochuli, Large-scale Chromatography of Recombinant Proteins, J. Chromatogr., 1988, 444, 293.
3. Y. C. Sasaki, Y. Suzuki and T. Ishibashi, Fluorescent X-ray Interference from a Protein Monolayer, Science, 1994, 263, 62.
4. G. B. Sigal, C. Bamdad, A. Barberis, J. Strominger and G. M. Whitesides, A Self-assembled Monolayer for the Binding and Study of Histidine-tagged Proteins by Surface Plasmon Resonance, Anal. Chem., 1996, 68, 490.
5. E. L. Schmid, T. A. Keller, Z. Dienes and H. Vogel, Reversible Oriented Surface Immobilization of Functional Proteins on Oxide Surface, Anal. Chem., 1997, 69, 1979.
6. R. Yasuda, H. Noji, K. Kinosita, and M. Yoshida, F1-ATPase is a Highly Efficient Molecular Motor that Rotates with Discrete 120°Steps, Cell, 1998, 93, 1117.
Handling and storage condition
| Appearance: | White to pale yellowish white powder |
|---|---|
| Purity (HPLC): | ≧ 97.0 % |
| Solubility in water: | To pass test (clear, colorless to slightly yellow) |
| IR spectrum: | Authentic |
| NMR spectrum: | Authentic |
| Ambient temperature |











